How can quality consistency be assured? What are the most important certifications? How long should the manufacturing and delivery process take? And which factories can be trusted to provide large quantities of sterile gauze pads without surprises? These are the kinds of questions that medical procurement professionals face on a daily basis, but the answers are often fragmented and found in a variety of different places.
The end result is uncertainty, and uncertainty is risk, particularly when it comes to high-volume consumable products like sterile gauze pads that are used in every hospital and every emergency situation.
This article pulls all of these answers together in one place, including the best manufacturers in China, the entire manufacturing process, and the sourcing standards that determine the best sterile gauze pads partners for 2026.
Key highlights
- The best quality and on-time delivery of sterile gauze pads come from manufacturers with more than 25 years of experience and exports to 70+ countries such as BKAMED, Winner, and Kingphar.
- ISO 13485, CE MDR, and FDA compliance and certification are the best predictors of a factory’s capability to provide compliant and audit-ready sterile gauze pads without customs holdups.
- Vertically integrated manufacturers who manage everything from cotton weaving to final sterilization provide better purity, absorbency, and quality of sterile gauze pads.
- The best factories are those that are open to physical audits and allow actual sample testing for absorbency, linting, and SAL 10⁻⁶ for large volume orders of sterile gauze pads.
Top 10 Sterile Gauze Pads Manufacturers in China
China dominates the industry in the production of vital medical supplies, with companies excelling in the production of sterile gauze pads that meet international standards for the treatment and healing of wounds and surgical procedures.
Let’s look at the top contenders and their unique contributions in the vital medical supplies industry.
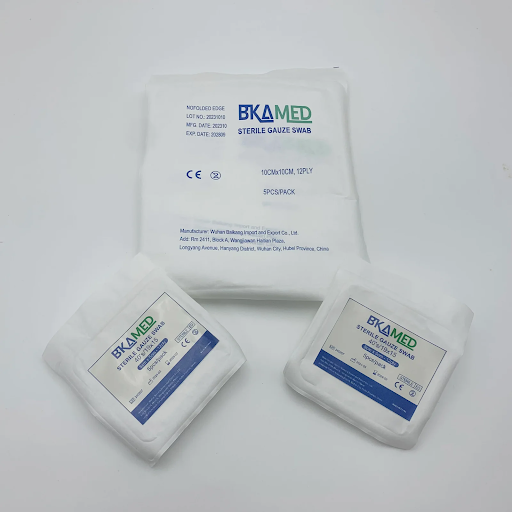
BKAMED started operations over 30 years ago as a specialized producer of medical disposables. The company, headquartered in Wuhan, Hubei Province, has multiple production bases distributed across Huanggang, Wuhan, and Xiantao. This helps the company to manufacture products in a streamlined fashion and respond promptly to the demands of its clients in the competitive medical disposables market.
The company exports its products to more than 70 countries, catering to the medical demands of Europe, Asia, North America, South America, Africa, and Oceania. The presence of strong certifications like ISO 13485, CE, and FDA approval ensures that the company is in compliance with global regulations. Today, all these elements have come together to make BKAMED a reliable partner for bulk medical textile requirements.
In the specific category of sterile gauze pads, BKAMED provides trusted alternatives such as 4×4 gauze pads, which emphasize maximum absorbency and a lint-free finish. These products are subjected to highly advanced bleaching and sterilization techniques to satisfy the stringent demands of operation theaters and emergency rooms. The company also manufactures other supporting products such as gauze rolls, variants of lap sponge, bandages, non-woven dressings, surgical tapes, and comprehensive first aid kits.
OEM and ODM services enable customized specifications according to specific hospital requirements. Modern hospitals process millions of units annually while using eco-friendly materials wherever possible. This helps ensure a steady supply chain without sacrificing sustainability.
BKAMED’s logistics system also helps with rapid turnaround, often shipping directly to major international ports within days. Healthcare buyers appreciate this reliability when managing inventory for large-volume hospitals.
Pros:
- Over 30 years experience ensures quality in sterile gauze pads.
- Exports to over 70 countries offer wide market reach for healthcare facilities.
- ISO and CE certifications ensure adherence to international standards.
- OEM/ODM services enable customized solutions for specific requirements.
- Eco-friendly production meets the needs of sustainability-conscious buyers.
Cons:
- The company’s main disadvantage is its lack of emphasis on advanced wound care, focusing only on basic sterile gauze pads.
- The company’s production depends on locations in Hubei Province, and disruptions in the area could impact its operations.
Winner Medical
Winner Medical was founded in 1991, based in Shenzhen, Guangdong, China, with a complete industrial chain of medical supplies.
The exports cover about 110 countries around the world, including Europe, Asia, America, Africa, and the Middle East.
FDA, CE, and ISO marks guarantee the global presence.
The company is a professional supplier of sterile gauze pads, which include gauze 4×4 pads for wound management and absorption.
The sterile gauze pads are sterilized and packaged for clinical use. The range of products includes wound management products, surgical products, alcohol prep pads, protective products, and advanced dressings.
Investment in R&D helps in the development of green materials, thus reducing the environmental impact.
The company provides tailored O.R. solution sets with sterile gauze pads integrated into the procedure sets.
The scale of the operation helps in providing competitive prices and long-term cooperation.
Pros:
- The industrial chain from raw materials ensures control over the quality of the sterile gauze pads.
- Exporting to 110 countries implies that there are extensive distribution channels for the organization’s products.
- High-tech automation ensures the efficiency of production processes.
- Emphasis on sustainability means that the organization’s services are in demand in the eco-conscious healthcare industry.
- High level of research and development ensures the organization’s innovative capabilities in the production of wound care products.
Cons:
- Increased focus on disposable products could imply that the organization does not specialize in the production of reusable sterile gauze pads.
- High level of production could imply that the organization responds slowly to customization demands.
Zhende Medical
It was founded in 1994 as a listed company, specializing in R&D of dressings and infection control products, located in Shaoxing, Zhejiang Province.
Zhende serves nearly 10,000 institutions across 73 countries, including Europe, the Middle East, Africa, Asia, and Russia. CE, ISO, and FDA certificates ensure smooth exports.
It offers sterile gauze pads, which include sterile gauze sponges, used for absorbent coverings in different plies.
The company also offers a range of products, including woven gauze sponges, bandages, surgical gowns, wound dressings, ostomy, and pressure therapy products.
It ensures sterility through modern automated production lines for sterile gauze pads.
Zhende partners globally, ensuring a wide reach through its ready-to-use kits, which include sterile gauze pads, with biocompatibility testing for safety in all applications.
Pros:
- The listed status ensures financial security for continuous R&D for sterile gauze pads.
- It serves 73 countries, focusing on institutional markets.
- Product offerings are diverse, catering to all healthcare needs.
- Infection control focus adds to product reliability.
- Custom packs simplify procurement.
Cons:
- Product variety may distract from innovations in core products, i.e., sterile gauze pads
Allmed Medical Products
Allmed was founded in 1997. The company is based in Shenzhen and producing in Hubei Province, and has been the biggest medical dressing export enterprise in China for the last 17 years.
Allmed has been delivering products to more than 70 countries on six continents, supported by ISO, CE, and FDA certificates.
Allmed medical supplies include sterile gauze pads, which include woven gauze sponges 4×4 sterile, which are breathable surgical prep pads. These pads include x-ray detectable varieties for added security.
The traditional range includes swabs, abdominal pads, and bandages, while the advanced range includes hydrocolloid dressings and foam dressings.
The intelligent production line guarantees the consistency of sterile gauze pads. Its ESG strategy includes initiatives on sustainable sourcing.
The customization range includes sizes and packaging, which are flexible for B2B businesses. Allmed’s strong export performance reflects the trust of the market.
Pros:
- High export ranking for 17 years ensures reliability in the quality of sterile gauze pads.
- Operates in 70 countries across the globe, covering all the continents.
- Intelligent manufacturing ensures efficiency in the production process.
- Emphasis on ESG ensures that the current procurement standards are met.
- Layout from spinning to sterilization ensures quality control in the entire chain.
Cons:
- High export orientation may cause the company to overlook the nuances in the domestic market.
- High variety in the products may cause the company to lose focus in the procurement process.
Hubei Qianjiang Kingphar Medical Material
Kingphar Medical started operations in 1989 in Qianjiang, Hubei Province, as a completely vertically integrated enterprise from cotton weaving to finished products. The company’s “Pure Oxygen” bleaching technology provides materials that are very white and pure. This vertical integration helps with cost savings and material purity.
Exports are made to over 80 countries, with major markets in North America, Europe, and Asia. ISO and CE certifications are the foundation of its international business. Its large production capacity, with 800 workers and more than 1,000 machines, can handle large OEM orders.
Sterile gauze pads have excellent absorbency, often combined with large bandages for trauma and post-operative use. Products include gauze rolls, swabs, lap sponge products, non-woven products, first aid kits, and infection control materials. Clean room conditions are maintained to keep products sterile throughout production.
Environmentally friendly versions are increasingly popular with progressive healthcare systems. Kingphar’s OEM partnerships with major brands worldwide demonstrate the trust developed over the past 30 years. Buyers appreciate the combination of economies of scale and flexibility that come with Kingphar’s experience.
Pros:
- More than 35 years of experience in the production of sterile gauze pads.
- Exporting to more than 80 countries ensures easy availability in the market.
- Having control over the weaving and bleaching process ensures quality control from the beginning.
- Having a large workforce ensures the production of large quantities.
- Having OEM business relationships ensures the building of strong business-to-business relationships.
Cons:
- Having the business located in the Hubei region may pose logistical problems.
Jiangsu Jianerkang Medical
Jianerkang Medical began operations in 1999 in Changzhou, Jiangsu Province, as a high-tech enterprise specializing in medical dressing products. The company’s large-scale purification workshops and automated production lines facilitate precise production. R&D work aims to enhance comfort and healing effects.
Products are distributed to global healthcare markets, especially in Europe and the Americas. CE, ISO, and FDA certifications allow for wide-scale compliance. The company’s over 100 product types cover a broad range of healthcare needs.
Sterile gauze pads come with non-adhesive gauze variants, which cause less irritation during dressing changes. These products can be combined with sponges, alcohol swabs, cotton products, and comprehensive dressing kits. The focus on non-irritating materials makes them ideal for sensitive skin use.
Modern production facilities strictly control environmental conditions to ensure product quality. Sales teams worldwide coordinate with distributors to provide for immediate needs. Healthcare institutions commonly choose Jianerkang Medical for its combination of innovation and reliable supply.
Pros:
- High-tech integration promotes the development of sterile gauze pads.
- Exports to key areas like Europe and Americas.
- Large variety of over 100 products.
- Large clean workshops promote compliance in terms of sterility.
- Emphasis on R&D promotes improvement.
Cons:
- May be limiting in terms of the scope of surgeries.
Suzhou Sunmed
Suzhou Sunmed has been in business since 1993 and has several bases in Jiangsu, Zhejiang, and Anhui Provinces. This helps to ensure business continuity despite local disruptions. The company’s 30 years of experience are in the clinical and consumer healthcare markets.
Exports are made to over 60 countries, including Europe, Southeast Asia, Africa, and Oceania. CE and FDA certification helps to enter tough markets. Sunmed’s production facilities are GMP compliant to ensure consistent production.
Sterile gauze pads are a part of a full line of products for wound care, including bandages, tapes, and cotton products. The company also distributes sports medical products and first aid kits. OEM services enable customization.
Recent product lines include kinesiology and cohesive bandages. This indicates continuous innovation. Purchasers in hospital groups appreciate the convenience of buying several product lines from one trusted supplier. Sunmed’s focus on every product batch helps to gain long-term trust.
Pros:
- Multiple production centers will improve flexibility in terms of supply of sterile gauze pads.
- Exporting to over 60 countries will cover a wide geographical area.
- CE and FDA certificates will ensure regulatory compliance.
- Offering a wide range of products, including first aid and sports,
Cons
- Diverse products offerings might dilute focus on sterile gauze pads.
Yangzhou Goldenwell Medical Devices
Goldenwell Medical began operations in 1988 in Yangzhou, Jiangsu Province, and is currently one of the largest medical device suppliers in China. The company has its own export licenses and offers favorable pricing for different product lines. Almost four decades of experience have honed their manufacturing skills.
Their products are distributed to markets all over the world, with major sales in North America, South America, and Europe. The company’s diversified product line allows facilities to make one-stop purchases. Their quality systems focus on performance and affordability.
Sterile gauze pads are fully compatible with dressing sets and surgical kits. They also produce syringes, infusion sets, tapes, bandages, and cotton products. This allows hospitals to acquire products from a single supplier without compromising quality.
Customized OEM services allow brands to create proprietary designs. Long-term partnerships with global distributors are a testament to the company’s stability. Experts in procurement services frequently mention the company’s capacity to provide value for money.
Pros:
- Almost 4 decades of business operations enhance the know-how of sterile gauze pads.
- Exports all over the world with their own licenses facilitate trade.
- Competitive prices attract bulk buyers.
- Offering OEM services facilitates customized manufacturing.
- A wide range of disposables meets the requirements of hospitals.
Cons:
- Concentrating on disposables may not focus on the latest sterile gauze pads technology.
Topwin (Hubei) Medical Products
Topwin started business in 1999 from its Wuhan, Hubei Province headquarters and currently has three large-scale production facilities. The company is dedicated to the production of medical gauze and personal protection products. A staff of about 600 enables large-scale production on an efficient scale.
Exports are made to over 20 countries, largely in Europe, the Middle East, and Asia. CE and ISO certifications guarantee that the products will live up to the buyer’s requirements. The end-to-end textile to sterilization process ensures strict quality control.
Sterile gauze pads come in hemostatic and bulk packs, depending on intended use. Swabs, elastic bandages, non-woven gowns, masks, and surgical accessories are also part of the product line. OEM services are provided to help global brands adapt to market requirements.
Ongoing R&D efforts ensure that the product line is constantly updated. Topwin has its own teams that work directly with clients to develop product specifications. This approach has helped Topwin establish itself among mid-sized healthcare systems.
Pros:
- Three big bases help scale up production of sterile gauze pads.
- Exporting to 20+ countries, with increasing presence.
- CE and ISO help ensure quality assurance.
- OEM expertise helps form partnerships.
- Over 25 years, refine our manufacturing processes.
Cons:
- Fewer countries for export compared to peers.
Nantong Jianan Medical Products
Jianan Medical started its operations in 1999 in Nantong, Jiangsu Province, with a specific emphasis on infection protection and wound care. The company’s clean workshops and standardized production ensure consistent quality. Its comprehensive production and export chain facilitates smooth international business.
The products are distributed in more than 20 countries in Europe, the Middle East, Africa, Asia, and Russia. CE, ISO 13485, and FDA certifications ensure easy approval. The company receives appreciation for its consistent quality, which satisfies the demands of medical practitioners.
Sterile gauze pads consist of large gauze pads, which are used for extensive coverage in trauma and surgical procedures. The product range includes rolls, bandages, swabs, abdominal pads, cotton products, masks, and non-woven products. The range meets both general and specialized demands.
The international medical community appreciates the company’s dedication to patient safety. Consistent production and good service have created a strong reputation among distributors. Jianan is continually upgrading its capacity to meet the increasing demand in emerging countries.
Pros:
- Specialization in protection increases the safety of sterile gauze pads.
- Exports to 20+ regions and has the required certification.
- Complete system from production to export.
- Focus on wound care meets specific demands.
- More than 25 years of production guarantees quality output.
Cons:
- A smaller scale may limit the potential of high-volume production.
What Are Sterile Gauze Pads?

Sterile gauze pads or sometimes known as sterile gauze swabs, are absorbent textile dressings made from purified fibers, most commonly cotton, that are sterilized to remove all living microorganisms. They are generally classified as disposable wound dressings that are designed to be used only once.
The use of these supplies is quite simple: to absorb fluids, protect the tissues, and maintain a clean environment between the wound and the outside world. As a medical device, these supplies are subject to a number of regulations that require them to be safe, clean, and packaged properly before distribution.
So, the most precise answer to the query of what are gauze pads would be that these are layers of porous dressings that are designed to manage fluid and cover wounds for a short period of time.
The weaving or bonding also allows the fluid to be drawn away from the wound bed. At the same time, air is also present to ensure that the wound is not subjected to maceration. This is the reason why sterile gauze pads continue to endure despite the availability of other advanced dressings.
Difference Between Sterile and Non-Sterile Gauze
Non-sterile gauze pads are often used for general purposes like cleaning or procedures where the chances of infection are less. These are often bulk-packed and are not subjected to sterilization.
In contrast, sterile gauze pads are processed through a validated sterilization method, and they are individually or small-pack wrapped. The wrapping maintains sterility until the moment of use. In operating rooms, emergency rooms, and invasive procedures, this difference is non-negotiable. The use of non-sterile materials in these environments will increase infection rates and expose facilities to potential clinical and legal issues.
From a procurement standpoint, this combination of materials in a tender will cause confusion and result in rejected shipments. Specifications must be clear on sterile, non-sterile, packaging, and intended use. Such clarity will enable accurate quotations from suppliers and compliance with hospital protocols.
How Sterility Is Maintained in Packaging and Use
Sterility is not only a matter of process, but it is also a matter of protection. Once sterile gauze pads are sterilized, they are moved to a controlled environment for packaging. Materials such as medical-grade paper, film, or peel pouches are used as a microbial barrier, yet they allow sterilizing agents to penetrate during processing.
Seals are also validated for integrity, which ensures that there is no contamination during transport and storage. The lot numbers and expiration dates are also added for traceability. As for the application, the clinical staff opens the package just prior to application, which reduces the overall time for the package. All these design elements, no matter how small they may seem, play an important role in maintaining sterility.
SAL (Sterility Assurance Level)
Sterility in the healthcare industry, in the context of manufacturing, is represented by the Sterility Assurance Level, commonly represented by the abbreviation SAL. The SAL for most sterile gauze pads is 10⁻⁶. The definition implies that the probability of the survival of a single microorganism during the sterilization process is less than once in a million.
Though the math may seem complicated, the idea is simple. The SAL ensures that the procurement teams are aware that the requirements for all batches are met, and the data for the same is available with the manufacturer in the form of biological indicators, validation, and monitoring data, which may be required during audits and supplier qualification.
Core Functional Roles in Clinical Settings

Absorption
Gauze, at its most fundamental level, is an absorptive material. The woven nature of the material draws fluids away from the surface, keeping the area clean and easy to assess. The multiple layers provide more absorptive capacity without adding bulk. As a means of absorption, it is extremely effective for short-term procedures.
Protection Against Contamination
After it has been applied, the sterile pads of gauze provide a protective mechanism that separates the wound from the outside world. The presence of dust, friction from clothing, and accidental contact with the outside world are all greatly diminished. This aspect of the gauze, in particular, becomes extremely valuable in the transportation of patients and in highly trafficked areas, such as emergency rooms.
Supporting Primary and Secondary Dressings
Gauze, in the majority of cases, serves either as the primary dressing in contact with the wound or as the secondary absorptive material applied over the top of the specialized dressing. The gauze provides support and stabilization for the dressing, as well as an even distribution of pressure. The non-adhesive nature of the material has become the preferred choice in the majority of cases, as it reduces trauma on the patient during the removal process.
Hemostasis Support
For minor bleeding or post-procedure maintenance, pressure in combination with absorbency is effective in managing the flow of blood. Stacked sterile gauze pads provide this support. The pads are economical enough to be changed often, which is usually required in the first hours of surgery.
Wound Packing vs Wound Covering
Medical practitioners use gauze pads in different ways depending on the wound. Shallow wounds require covering, while deeper cavities require packing. The former helps protect the wound surface and manage moisture, while the latter helps in drainage and prevents premature healing. The same base material is used for both purposes with little modification.
Shapes, Sizes, and Formats
There is little chance of hospitals using only a single size of gauze pads. The most commonly available gauze pads are the standardized ones, i.e., 4×4 gauze pads. The smaller gauze pads are required for delicate procedures, while larger gauze pads are required for more exudate or larger surgical sites.
Common forms include:
- Flat, multi-ply squares
- Pre-folded pads for rapid placement
- Strips or rolls for packing
- Bulk packs or sterile singles
Hospitals also acquire procedure kits that contain sterile pads with tape and secondary dressings. These make the preparation process easier and save valuable time in busy departments.
For heavy-absorbency procedures, healthcare teams may use thicker pads, also known as sterile gauze sponges. For surgical procedures, larger absorbent pads, such as a lap sponge, are used for fluid control and counting procedures. While these are similar in nature, they are used for more specific purposes than the standard pads.
Material Types
Woven Cotton Gauze
Traditional gauze sponges are made of woven cotton yarns, creating a lattice structure that provides strength and absorbency. Many clinicians favor the traditional woven gauze sponges because of their ability to maintain their shape during use. The natural softness of cotton also makes it comfortable for the patient.
Non-Woven Rayon/Cotton Blends
Non-woven sponges are made of bonded fibers instead of the traditional woven material. These sponges are often less prone to lint and can be produced with higher uniformity. These sponges are lighter and, in some cases, may be less expensive for large-volume facilities. In some tender documents, these sponges may be beneficial by reducing waste and increasing density.
Specialty Materials
Some sponges may be required for specific tasks, requiring additional material properties. These sponges may be made with different materials and coatings to meet the requirements of these tasks. In standardized procedures, many tender documents favor the traditional woven gauze sponges 4×4 sterile.
Performance Characteristics That Matter to Procurement
From a sourcing perspective, performance data is far more important to a purchasing decision than marketing information. Key performance attributes are:
- Absorbency rating (mL per pad)
- Tensile strength
- Linting and fiber shedding
- Conformability
- Breathability
These attributes are critical to the performance of a sterile gauze pad, which must perform reliably for thousands of daily uses. Fibers may break during removal, and linting may cause wound contamination. Breathability, on the other hand, is important for creating a proper wound-healing environment. Testing samples against these attributes will allow for a fair comparison of vendors.
Where Sterile Gauze Pads Are Used
The demand pattern covers almost all the care environments. In the operating room suites, the sterile gauze pads are used to help manage fluids and dress wounds. In the emergency room suites, the sterile gauze pads are also essential to quickly stop bleeding and cover wounds. In the wound care centers, the sterile gauze pads are needed daily to regularly redress wounds.
In the field hospitals and the mobile clinic environments, the sterile gauze pads are preferred because of their portability. In humanitarian organizations, the bulk cartons are needed because the sterile gauze pads are easy to store and can be adapted to different kinds of cases. The ease of use makes training easy.
Differences vs Other Wound Care Products
Sterile vs Non-Sterile

As previously explained, the sterile status of gauze pads is a key factor in wound care products. Sterile gauze pads are required in infection control procedures, while non-sterile gauze pads are required in secondary or non-critical procedures.
Gauze vs Advanced Wound Dressings
Advanced dressings include alginate, foam, and hydrocolloid dressings. However, they are more costly and limited in use compared to gauze pads.
When Gauze Pads Are Preferred
Gauze is often chosen when:
- Frequent dressing changes are required
- Costs must be low
- Versatility is a requirement
- Training levels vary
In the above conditions, the reliability of sterile gauze pads makes them the preferred option. Even in facilities where advanced dressings are available, gauze pads are in high demand because of the gaps they cover in the dressings.
What Defines a High-Quality Sterile Gauze Pad

In bulk medical procurement, a sterile gauze pad is not merely a commodity item, but rather a performance tool that has a direct impact on infection rates, nursing efficiency, and treatment costs.
While many vendors describe their gauze pads as “medical grade” or “hospital quality,” this information has limited practical value to a purchasing manager. What matters most is performance, as measured by factors such as absorbency speed, purity of fibers, strength, sterility, and packaging.
In order to understand quality, one must look beyond surface qualities and price per carton to consider how a given item will perform across tens of thousands, or even millions, of uses.
The following sections outline the technical and operational considerations that distinguish a quality sterile gauze pad from commodity-grade products.
Core technical specifications
From the institutional perspective, sterile gauze pads are rated based on objective specifications rather than marketing information. These specifications define the ability of the product to work in the operating rooms, emergency rooms, and clinics in the field.
Cotton purity
Cotton purity is a key factor in the safety and efficacy of the product. High-quality sterile gauze pads contain 100% bleached cotton, which is free from seeds, synthetic materials, oils, and other foreign matter.
Impure materials may compromise the ability of the product to absorb fluids, which may also cause linting or shedding of fibers in the wound. For the hospital, this may mean a potential infection risk and compromised healing. As a result, the hospital’s procurement department seeks vendors who can provide raw materials test reports.
Thread Count
Thread count is the density of the weave.
More thread count means:
- More structural strength
- More resistance to fraying
- More even fluid distribution
Lower thread counts may be cost-effective but will also be prone to collapsing or deformation when wet. For surgical and post-surgical recovery, consistency is often the best value, making higher thread counts the institutional norm.
Ply Thickness
Ply refers to the number of layers of fabric compressed into a pad (most often 8-ply, 12-ply, or 16-ply).
More plies mean:
- More absorption
- More cushioning
- More protection
More plies also mean a larger product size and higher materials cost. Purchasing decisions often weigh the potential wound severity against the efficiency of storage and shipping.
Absorbency Rate
Absorbency is not just about overall capacity, as speed of absorbency is also important. Gauze needs to be able to quickly draw blood or exudate away from the wound surface.
Laboratory tests for absorbency rates, rather than visual evaluation, offer a more scientific measure of a product’s effectiveness (in seconds or ml/g).
Top-performing products help minimize risks as well as nursing time.
Sterility Assurance Level (SAL)
Sterility Assurance Level (SAL) is a measure of the probability of a viable microorganism surviving a sterilization process.
For surgical-grade sterile gauze pads, a standard SAL of 10⁻⁶, or a one-in-one-million probability of a microorganism surviving, is typically recommended.
Suppliers of gauze pads must be able to demonstrate:
- Sterilization method (e.g., EO, gamma, steam)
- Batch testing
- Traceability
Without this, even a highly effective gauze pad will not be suitable for sale in a regulated healthcare environment.
Packaging formats for institutional use
The packaging has a direct impact on the operation in terms of workflow and waste management.
The different packaging formats for institutional use are:
- Individually sterile wrapped pads
- Peel packs
- Multi-count sterile pouches
- Bulk non-sterile cartons for internal sterilization
The individually wrapped pads provide the highest infection control but have a higher waste management cost. In hospitals, multi-count sterile packs provide the best overall solution, offering the best balance of accessibility and infection control.
The purchase decision is based on:
- Shelf life
- Carton size efficiency
- Labeling clarity
- Lot traceability
- Storage ease
The packaging design helps in reducing the handling time of the nursing staff, which in turn reduces the operation costs more than the price difference of the products.
Performance differences: woven vs non-woven
Material construction plays a vital role in determining performance characteristics.
Woven gauze pads are constructed using traditional interlaced cotton threads. They are:
- Durable
- Familiar to clinicians
- Cost-effective
- Suitable for general wound care
However, they are prone to linting and may bond to the wound.
Non-woven gauze pads, constructed from compressed synthetic or blended fibers, possess:
- Less linting
- Smooth surface finish
- Higher fluid retention capacity
- Consistent material structure
Such properties are beneficial for surgical procedures.
In terms of sourcing, woven gauze pads are generally available at a lower unit price, whereas non-woven gauze pads are advantageous for minimizing wound-related complications and reducing dressing changes.
In a number of hospitals, a combination of non-woven and woven gauze pads is employed, with non-woven being utilized for critical care and woven for routine care.
Cost vs. performance trade-offs
Decisions regarding procurement should not be based solely on the price per pad. Instead, it should be based on the cost per treatment.
A lower-cost gauze might seem to be a good value, but there are hidden costs:
- More frequent dressing changes
- More time spent by nurses
- Product waste
- Infection-related complications
- More inventory usage
A high-performance sterile gauze pad might save costs in these areas, providing a longer-lasting product with better performance.
If a high-performance pad reduces dressing changes from three to two per day, it might save labor costs, even if it costs a bit more.
A wider view of these costs changes the perspective on gauze from a commodity to a strategic product.
As a result, many healthcare organizations have a process for evaluating suppliers based on performance testing, trial usage, and cost per case, rather than price per product.
The Complete Manufacturing Process of Sterile Gauze Pads
Under every box of sterile gauze pads, there is a process that is much more complicated than what the end buyer typically wants to know. While gauze may appear to be a simple cotton product, the process of creating the uniform quality required by hospitals and other end users requires precise control of the material, the fibers, the weaving, and the sterilization.
For procurement professionals, the process helps answer the question of why some suppliers are able to deliver uniform quality year after year, and others may be plagued by problems like lint, absorption, sterilization, and even rejection.
When it comes to bulk healthcare products, small problems in the manufacturing process can quickly turn into big problems in the operations process. That’s why serious manufacturers of sterile gauze pads think of them as medical devices, not textile products.
The sections below walk the reader through the process by which serious manufacturers of gauze pads ensure quality, even if the end buyer never wants to know the details.
Raw Cotton Sourcing and Inspection
The quality of the product begins from the fibers. If the raw cotton is of poor quality, then it is not possible to improve the final product.
Established factories source medical-grade, long-staple cotton from accredited farms or suppliers. This is because the longer staple fibers produce a stronger yarn, which has lower linting and higher absorbency. All these factors are crucial in the production of sterile gauze pads.
The raw cotton supplied to the factories typically undergoes the following inspection processes:
- Visual inspection of impurities
- Moisture content test
- Fiber length grading
- Contamination test, which includes dust, seeds, and other foreign particles
Poor-quality cotton can cause contamination, which may later be present in the final product, leading to infections in the wound.
Established factories discard poor-quality raw materials at the early stages rather than attempting to process the materials. This ensures the quality of the product and the reputation of the firm.
From the perspective of the firm, the suppliers who control the raw materials supplied to the factories experience very few quality-related problems.
Scouring and Bleaching
Raw cotton has waxes, oils, and pigments that are not only aesthetically unpleasing but also impair its ability to absorb. These must be removed for it to be viable as a medical gauze.
Scouring involves controlled washing and alkaline treatment to remove waxes, oils, and other substances. Bleaching follows to produce a clean, white appearance and enhance its capacity for absorbing liquids.
This process has a direct impact on:
- Absorbency speed
- Odor neutrality
- Cleanliness of fibers
- Compatibility with human skin
If scouring is inadequate, gauze will repel liquids rather than absorb them well.
In modern factories, this process is carefully controlled, with precise attention to chemical concentration, temperature, and washing cycles. This helps prevent any variation in gauze batches, which could manifest as differences in performance.
A well-executed process at this stage will yield a gauze that is soft, easy to absorb, and performs well.
Carding and Weaving
The cotton fibers, once cleaned, are arranged and bundled together to create a yarn, a process known as carding.
The size and consistency of the yarn are crucial to the production of a sterile gauze pad, and any irregularities in the yarn could create weaknesses or areas of non-absorption.
The weaving of the yarn into a fabric, or gauze, involves the use of a series of controlled parameters, including:
- Thread count
- Mesh size
- Tension
- Density
The parameters are controlled to create a fabric suitable for the production of a sterile gauze pad.
The weaving of the gauze could be loose or tight, and although a loose weave could save production costs, it could lead to fraying and lint, whereas a tight weave could make it more durable but slightly less flexible.
Manufacturers have standardized certain weaving configurations to meet the requirements for a sterile gauze pad, and the machines used to create these gauze fabrics are much more consistent in their production than manual or semi-automated systems.
Cutting and folding automation
Once the weaving process has been completed, the fabric is cut into specific sizes and folded into pads.
This process may seem straightforward, but cutting the fabric by hand often causes inconsistencies in terms of size, orientation, and ply layers. Even minor inconsistencies in the cutting process can lead to difficulties in the packaging or usage of the gauze pads.
Leading manufacturers utilize cutting and folding machines to ensure:
- Exactness in the size of the pads formed
- Accurate ply layers
- Clean cutting edges
- Minimal shedding of fibers
The use of automation in cutting and folding the gauze pads increases the overall speed of the process while minimizing the chances of contamination.
This process may be of interest to procurement teams who often wonder why some manufacturers provide pads in neat stacks while others may be of inconsistent size and poorly stacked.
Sterilization methods
Sterilization is a key process that differentiates gauze pads from non-sterile ones. Each batch of sterile gauze pads must achieve a validated sterility assurance level.
Various sterilization processes are applicable to different operational processes.
EO sterilization
Ethylene oxide sterilization employs gas penetration into packaging to destroy microorganisms at low temperatures.
Its advantages are:
- Excellent gas penetration into packaging
- Suitable for sealed packaging
- Material compatibility
However, ethylene oxide sterilization has a drawback:
Aeration time is required to remove ethylene oxide, increasing processing time
Despite longer processing times, ethylene oxide sterilization is popular for mass production of medical consumables because of its reliability.
Gamma radiation
Gamma sterilization employs gamma rays to destroy bacteria and spores.
Gamma radiation sterilization has several advantages:
- Fast processing time
- Deep penetration into material
- No chemical residues
This method is efficient for high-volume production of sterile gauze pads, especially when quick turnaround is needed.
However, gamma radiation sterilization has a drawback: Material degradation may result if gamma doses are excessive, making dose control crucial.
Gamma radiation sterilization is popular among many large exporters.
Steam Sterilization
Steam sterilization utilizes high pressure saturated steam.
It is:
- Cost-effective
- Chemical-free
- Environmentally friendly
However, the heat and humidity can cause deformation of packaging materials and are not suitable for pre-sealed sterile packaging.
Therefore, steam sterilization is more likely to be used internally within hospitals rather than for exporting products.
Each company will use different sterilization techniques depending on the type of product manufactured, cost structure, and target markets.
Cleanroom Packaging
Once the sterilization process is complete, the sterile gauze pads are protected from re-contamination.
Therefore, packaging occurs within a cleanroom environment where the air filtration, humidity, and particle levels are controlled.
Individuals undertaking the task will follow:
- Gowning
- Restricted access
- Sanitized workstations
- Minimum handling
Any small particles present in the air can contaminate the sterility of the packaging.
Therefore, the environment is constantly monitored to ensure sterility is maintained.
Cleanroom packaging is an indication that the company is more likely to follow international regulations.
Batch coding and traceability systems
Traceability has become a very important factor in the global supply chain in the health industry.
Every carton of sterile gauze pads must be traceable back to:
- Production date
- Raw material lot
- Sterilization batch
- Inspection records
Batch coding allows for quick recall in the event of a problem and facilitates audits in the European Union or the United States.
Without traceability, it is very difficult to manage risks for both the manufacturer and the buyer.
With the help of digital traceability systems, complete lifecycle documentation is possible, which is very beneficial to hospitals and NGOs that must comply with strict regulations.
Final quality inspection protocols
Finished sterile gauze pads must be inspected before they are shipped.
They are checked for:
- Absorbency
- Dimension accuracy
- Ply
- Visual defects
- Seal integrity
- Sterility
All these tests must be done to ensure consistency in the product.
Where these problems typically occur
These problems typically involve issues with loose threads, incomplete sterilization, or improper sealing and folding. These are often the results of rushed manufacturing or an uncontrolled process.
How serious manufacturers prevent contamination
Serious manufacturers attempt to prevent these issues through an automated process, employee training, and clean room environments with multi-stage inspection systems.
Certifications, Regulatory and Compliance Standards for 2026
As the global medical procurement landscape continues to evolve, the quality of the product is no longer the sole determining factor. Instead, the documentation, certifications, and regulatory requirements play the critical role of determining whether the goods are cleared by customs and reach the port warehouse quickly or are stuck in a warehouse.
This applies especially to sterile gauze pads, as they are classified as ‘regulated medical devices.’ This classification means there are stringent regulations, audits, and the provision of technical documents.
For hospitals, distributors, and NGOs, the process of compliance is not simply one of ticking boxes; it has serious implications for the smooth-running of operations and the provision of care.
Today, however, compliance with regulations also carries another important implication: it demonstrates the presence of systems, as opposed to ‘ad-hoc’ manufacturing.
The sections below discuss the fundamental requirements procurement teams should be aware of, provided by the supplier of sterile gauze pads, as we move into 2026 and beyond.
ISO 13485 medical device quality systems
International Organization for Standardization certification under ISO 13485 is the basic requirement for serious medical device manufacturers.
ISO 13485 is a set of rules that emphasizes the importance of a structured quality system rather than product testing.
For a sterile gauze pad, the key areas under ISO 13485 are:
- Raw materials
- Sterilization
- Traceability
- Corrective action
- Internal and external audits
Without ISO 13485, it is difficult to establish consistency.
From a procurement point of view, ISO 13485 certification is a prediction of the vendor’s reaction in a crisis situation. ISO 13485-certified factories tend to fix problems at the root cause rather than the symptoms, ensuring that problems do not recur.
ISO 13485 is often a non-negotiable requirement in large tenders because it reduces the risk of the entire supply chain.
FDA 510(k) or listing requirements
For products entering the U.S. market, there is regulatory control by the U.S. Food and Drug Administration.
For medical consumables, e.g., sterile gauze pads, listing or clearance by the FDA, depending on the product, is a regulatory requirement.
This ensures that the product meets regulatory expectations, e.g.,
- It meets safety standards
- It is similar to predicate devices
- It was made under a compliant quality system
- It was made by a registered facility
- It can be traced back to a registered facility
For international companies, being compliant with FDA regulations is a sign of being ready for a highly regulated environment in a global healthcare system. Even companies not in the U.S. would want to do business with companies registered with the FDA, as their documentation standards are strict and transparent.
CE MDR compliance
For exports to Europe, the European Commission has a specific framework under the European Union’s Medical Device Regulation, which is abbreviated as CE MDR.
According to the MDR, sterile gauze pads are subject to more stringent requirements compared to the previous directive system.
The requirements include:
- CE marking
- Technical documentation
- Clinical evaluation or literature support
- Notified body audits
- Unique Device Identification (UDI)
The MDR environment holds manufacturers accountable for the entire lifecycle rather than a point-in-time approval.
From a procurement team’s point of view, CE MDR compliance by a medical device manufacturer implies the ability of the firm to withstand audits and maintain discipline in the long term.
This minimizes the risk of any unplanned action by the medical device manufacturers, which may result in the termination of the supply contract.
WHO and NGO procurement standards
For humanitarian organizations and public health entities, the World Health Organization may be a resource for procurement standards.
These standards generally emphasize the safety, reliability, and appropriateness of the product for harsh environments.
For sterile gauze pads, these requirements may include:
- Clear labeling
- Long shelf life
- Robust packaging
- Resistance to heat and humidity
- Lot tracking
Field hospitals and disaster response operations require products withstanding transportation, storage, and extreme environmental conditions.
As such, it’s not unusual for NGOs to require additional documentation, especially if the supplier is familiar with WHO procurement standards.
This attention to real-world conditions, as opposed to hospital storage, can be critical in these environments.
Sterility validation documentation
Sterility is something you need to prove, rather than assume.
Sterile gauze pads manufacturers need to prove the sterility assurance level of their sterilization process.
Procurement teams need to expect the following:
- Sterilization validation reports
- Routine cycle records
- SAL confirmation
- Packaging integrity testing
- Revalidation schedules
Without this documentation, sterility is just a marketing promise rather than a scientific reality.
Good sterility documentation is something well-run factories keep well-organized, as audits and tender documents often ask for it.
In reality, good sterility documentation can be the difference between a good exporter and a small workshop unable to cope with international standards.
Environmental and sustainability regulations
Environmental regulations are now an integral part of supplier qualification, especially in Europe and North America.
New regulations are now looking at:
- Wastewater treatment
- Chemical handling
- Energy efficiency
- Responsible sourcing
- Packaging waste reduction
Sterile gauze pads, being disposable, may seem harmless, but the production process still has environmental implications.
Hospitals and NGOs are now looking for suppliers with sustainability initiatives in place.
It’s not just about the PR; it’s also about the law. Factories that are non-compliant are at risk of closure or export bans, which affects the supply chain.
So, sustainability initiatives are not just good PR; they also contribute to the reliability of the supply chain.
Common compliance gaps that delay imports
It’s not uncommon for delays in imports not being due to major non-compliance, but rather minor documentation issues.
Issues that commonly arise are:
- Expired certificates
- Mismatched labeling
- Lack of batch numbers
- Incomplete sterilization documents
- Incorrect HS codes
While the sterile gauze pads themselves are of acceptable quality, paperwork issues can bring clearance to a standstill for weeks.
Experienced exporters often use dedicated compliance teams to sort through these issues.
For the buyer, suppliers that regularly ship goods abroad are less likely to experience administrative problems.
Experience can count for a great deal.
Documents that procurement departments should always ask for
In most cases, the following documents are requested before the order is finalized:
- ISO 13485 certificate
- FDA registration or listing
- CE documents
- Documentation of sterilization validation
- Batch traceability samples
- Product specifications
- Packaging information
- Shelf life information
Requiring these documents beforehand can help ensure that the buyer is not surprised later on.
If the supplier can provide these documents readily and accurately, it is likely that the systems behind the manufacture of the sterile gauze pads are well-developed.
How Hospitals and NGOs Evaluate and Select Suppliers
The selection of a supplier for medical consumables is not a simple exercise of comparing prices. For hospitals, public health systems, and even humanitarian agencies, the selection of a supplier for medical consumables is a complex exercise.
The above statement assumes even greater significance in the selection of a supplier for sterile gauze pads, which are used daily in various operations and emergency situations.
The selection of a supplier for sterile gauze pads is done through a structured process, and the entire exercise involves documentation, auditing, and testing, and finally, risk assessment.
The point to note here is, the best partnerships are built on systems and evidence, and not on promises.
The sections below outline how a professional buyer selects a supplier for sterile gauze pads in a structured and disciplined manner.
Supplier qualification checklists
Organizations generally start with a formal qualification checklist. In this step, suppliers that are not able to meet the minimum technical or regulatory standards are eliminated.
The following are the most common categories that are included in the qualification checklist:
- Regulatory certifications
- Manufacturing capacity
- Product specifications
- Sterilization processes
- Export experience
- Financial condition
- Lead times
- After-sales services
In the case of sterile gauze pads, the checklist would include sterility validation, packaging integrity, and lot traceability.
Documentation is the first step to qualify the supplier. If the key documentation is missing, the supplier is eliminated at this stage to avoid wasting time on the supplier later on.
These qualification checklists are standardized documents that ensure all suppliers are evaluated based on the same criteria.
Factory audits and virtual inspections
After the initial qualification, the next step in the process for most companies is the factory audit.
Audits provide an opportunity for the procurement or quality teams at the companies that buy the sterile gauze pads to gain an in-depth look at how the sterile gauze pads are made, not just how they are described in the documentation.
Audits are conducted in the following areas:
- Cleanliness and layout
- Workflow separation (dirty and clean zones)
- Equipment maintenance
- Staff training
- Quality records
- Storage conditions
If travel for the audits is not feasible, virtual audits are also popular, using video and document-sharing technology.
While virtual audits are not the same as in-person audits, they still provide an indication of the level of maturity and openness in the operations. A factory that is open with their operations is likely more controlled than one that is not.
Sample testing methods
Prior to making a commitment to bulk purchases, a product sample is typically requested by the procurement department.
This process ensures that the sterile gauze pads perform well under real-world conditions.
While product specifications are provided in the lab, practical experience often differs from the theoretical.
Some of the tests conducted on the product sample are:
- Absorbency speed and capacity
- Tensile strength
- Linting or shedding of fibers
- Seal strength of the packaging
- Sterility indicators
Some institutions may also test the product under various conditions, such as fluid management during a procedure or under long-term storage conditions.
Other products, such as sterile gauze sponges, may also be used as a comparative test, particularly when considering compatibility with a particular set of dressings.
The objective is to establish whether the product has a consistent performance record, not only once but multiple times, before making a substantial financial commitment.
Risk scoring suppliers
Not all suppliers are the same in terms of the risks they pose.
For objective decision-making, organizations often use a system of risk scoring based on various quantifiable factors.
Some of the key factors in determining the risks associated with a particular supplier are:
- Regulatory compliance record
- Production capacity in relation to demand
- Geographical risks
- Past delivery record
- Financial stability
- Quality complaint record
For sterile gauze pads, the stockout risk is a key factor since the product has a daily consumption pattern in large quantities.
A low-cost supplier who has a poor delivery record may score lower than a slightly higher-cost supplier who has a good delivery record.
Negotiating MOQs and pricing
After technical suitability has been established, commercial negotiations will commence.
Minimum Order Quantities (MOQs) can play a significant role in determining purchasing strategies. Higher MOQs may result in lower costs, but they also result in higher inventory costs.
For Sterile Gauze Pads, high consumption rates will allow for bulk purchasing, but NGOs and smaller hospitals may still require flexible MOQs.
Negotiations will generally cover:
- Tiered pricing
- Volume discounts
- Mixed containers
- Private labeling
- Payment conditions
- Freight costs
- Duties and storage costs
In many cases, it is not just a matter of factory pricing. Freight, duties, and storage costs can significantly alter the actual costs. Pricing discussions are therefore typically undertaken on a total value basis rather than a headline price.
Contract structures
Formal contracts establish expectations and provide security for both parties.
This structure helps avoid misunderstandings and ensures that, in the event of issues, there are established remedies available. For medical supplies, this structure is especially relevant.
For contracts on sterile gauze pads, the structure may include:
- Product specifications and tolerances
- Quality acceptance criteria
- Delivery timetables
- Penalties for delays
- Replacement policies
- Regulatory requirements
- Confidentiality and labeling requirements
Audits and/or performance reviews, in some cases
Formal contracts eliminate the need for opinion-driven negotiations, replacing them with concrete requirements that are easier to resolve in the event of disagreements.
Long-term framework agreements
Instead of constantly placing orders for medical supplies, some institutions enter long-term contracts, also known as framework agreements.
This type of contract secures prices, pricing structures, and supply commitments for anywhere from one to three years or longer.
For contracts on sterile gauze pads, the long-term structure offers:
- Guaranteed pricing
- Guaranteed production slots
- Priority allocation in the event of shortages
- Ordering ease
Manufacturers also reap the benefits of better predictions of demand.
This stability, shared by the buyer and the supplier, often encourages cooperation and the discussion of continuous improvements, rather than renegotiation.
Long-term relationships are often the most effective procurement strategy in high-volume categories.
Real-world procurement workflow
In the real world, the process tends to follow a predictable pattern.
Here’s what the workflow might look like:
- Technical requirements defined
- Supplier prequalification issued
- Review of certifications
- Conduct audits
- Test samples
- Score risks
- Negotiate prices
- Sign contracts
- Monitor performance
At every phase, the uncertainty decreases. By the time orders are placed for sterile gauze pads, most of the risks have already been evaluated and documented.
This process helps the procurement staff justify the process internally and maintain consistency even if the staff changes.
Consistency breeds consistency.
Tenders vs. Direct Sourcing
Tenders and direct sourcing have different uses. Public hospitals and NGOs tend to make use of tenders, whereas private hospitals might prefer to make direct procurements.
Tenders promote transparency and competition. They standardize the requirements and encourage multiple tenders, thus ensuring a fair and reasonable price.
On the other hand, direct sourcing provides a sense of urgency and flexibility. It is best to make direct procurements when there are trusted suppliers of sterile gauze pads who have shown good performance.
Both have their own benefits although tenders are best for public hospitals, whereas direct procurements are best for private hospitals.
Supply Chain, Logistics, and Import Considerations

However, sourcing quality products is only half of the picture. Even a quality manufacturer will be unable to sustain a hospital’s clinical operations if their products are delivered late, held up at customs, or damaged during transportation.
In the case of high-volume consumable products like sterile gauze pads, logistics performance is a key factor because they are used daily in operating rooms, emergency rooms, and outreach programs.
Procurement teams consider supply chain planning a vital part of their overall risk management strategy. A predictable delivery schedule, transportation, and customs procedures are as important as price.
Let’s consider how hospitals, distributors, and NGOs organize their logistics to deliver a constant stream of sterile gauze pads from factory to facility.
Lead Times and Production Planning
The lead time starts much before the goods are shipped out of the factory.
The production process for sterile gauze pads includes cotton processing, weaving or non-woven fabric forming, cutting, sterilization, packaging, and inspection.
The components of lead time are:
- Sourcing of raw materials
- Manufacturing time
- Sterilization time
- Quality checks
- Packaging
- Export documentation
- Transit time
Manufacturing time is typically quoted for a period of 30-45 days, but it could be longer during peak seasons.
A seasoned buyer, however, uses a rolling forecast and blanket purchase orders to place production well in advance instead of waiting for stocks to run out.
This ensures a stable production schedule for the factory and prevents costly and urgent shipments later.
Container optimization strategies
Freight costs are often an invisible drain on the bottom line, especially for lightweight, bulky products.
Sterile gauze pads, for example, are often more bulky than they are heavy, causing shipments to “cube out” long before they “weigh out.” The result: wasted valuable container space.
To optimize container fill, suppliers and buyers usually:
- Standardize carton sizes
- Use vacuum or compressed packaging wherever possible
- Stack pallets to maximum height
- Optimize the mix of different SKUs
- Consolidate several orders in one container
Small improvements in packaging can add up to big improvements in container fill rates.
Higher container fill rates not only save per unit freight costs, they also reduce the environmental impact at the same time.
For large institutional buyers, these savings add up fast, especially with multiple shipments per year.
Air vs Sea Freight
Cost and reliability are influenced by the mode of transport used.
Sea freight is the most commonly used mode for the transportation of sterile gauze pads, as the quantity is large and the value per unit is low. Sea freight has the lowest cost per unit, but the lead time for transportation is long.
On the other hand, air freight has the shortest lead time, which means that the cost per unit is high. Air freight is used in emergency cases, launch orders, and in the event of an unexpected shortage.
Trade-offs:
- Sea Freight: Low cost, long lead time
- Air Freight: High cost, short lead time
Most procurement teams use both modes in combination:
Sea freight for regular replenishment and air freight for safety quantities in emergency cases.
Customs clearance challenges
Delays are common for shipments crossing borders, especially for medical devices that are subject to regulations.
Sterile gauze pads, being medical devices, are subject to specific requirements in different countries, and the relevant authorities may demand additional documents apart from the regular commercial invoice used for other shipments.
The requirements that are usually demanded for the clearance of the medical devices include:
- Certificate of conformity
- Sterility validation summary
- Product registration numbers
- HS codes
- Compliance with labeling requirements
- Country of origin
Without the relevant documents, the cargo may be stuck in transit for days or weeks. Experienced exporters ensure that all the documents are ready prior to shipping the medical devices and work with customs brokers who are familiar with healthcare shipments.
Cold chain or sterile handling requirements
Sterile gauze pads, unlike pharmaceuticals, do not require refrigeration. However, sterile handling is still required.
The packaging of the gauze pads is an important factor in this regard.
The packaging of the gauze pads is usually in the form of sealed pouches, medical-grade paper, or peel packs, which retain sterility as long as the package remains sealed. The cartons must be resistant to moisture, dust, and crushing.
The best practices in this regard include:
- Dry storage conditions
- Protection from extreme heat
- Palletization to avoid floor contact
- Careful handling to avoid damage to the package
Although the gauze pads do not require refrigeration, damage to the package can still be a problem.
Regional warehousing strategies
Some organizations use a risk reduction approach where inventory is held closer to the end users.
Instead of shipping directly from the factory each time the inventory is needed, the buyer could use a warehousing or logistics company to stock up on the sterile gauze pads.
There are several advantages to this approach:
- Faster delivery times
- Reduced costs associated with emergency shipments
- Inventory to cover unexpected surges in use
- Simplified customs procedures
Regional warehousing is especially important to large healthcare systems or NGO networks with several different facilities.
By reducing the last mile of the delivery process, the hospitals can gain flexibility without being beholden to international shipping schedules.
How to prevent stockouts in humanitarian operations
Humanitarian missions are often conducted in very remote or unstable environments where inventory can arrive at unpredictable times.
To mitigate this risk, NGOs usually:
- Keep higher levels of safety stock on hand
- Pre-position supplies in multiple locations
- Use conservative consumption forecasts
- Diversify the supply base
- Blend sea shipments with a periodic air component
Redundancy is a design principle.
It may cost a little more in inventory costs, but it pays off in the clinic when the roads close, the ports slow down, or the crisis suddenly spikes demand.
In humanitarian logistics, resilience is more important than efficiency.
Sustainability, Ethical Sourcing, and ESG Expectations
In the past, the evaluation of medical consumables has been based on price and performance. Today, sustainability and ethical sourcing are also important criteria for selecting suppliers, especially for public hospitals, multilateral agencies, and large NGOs.
Products like sterile gauze pads are often straightforward and uncomplicated. But the manufacture of such consumables involves various stages like the cultivation of cotton, processing of cotton into usable form, packaging materials, transportation of the finished product from the country of manufacture to the consuming country, and so on. Each of these stages has its own environmental impact.
Today, the focus is no longer just on the finished product. The focus is also on how the finished product has been made, under what conditions it has been made, and with what kind of cost to the environment and society as a whole.
Here’s the thing: sustainability is no longer just a brand story. Today, sustainability is a compliance and risk management imperative linked to funding models, tendering processes, and public accountability.
The following are the areas where the expectations are rising the fastest:
Organic or responsibly sourced cotton

Buyers increasingly require suppliers to prove:
- Traceability of the cotton source
- Reduced chemical use
- Water-efficient cultivation
- Good labor practices
While not all responsible sourcing requires organic certification, it promotes confidence.
Companies with direct control over their supply chain generally offer better quality and lower risk.
Wastewater and bleaching chemicals
Scouring and bleaching steps involve wastewater and chemical by-products. If not properly managed, these chemicals can pollute local waterways.This poses environmental and legal risks, hindering the manufacturing process.
For the production of sterile gauze pads, responsible manufacturers:
- Reduce wastewater and chemical by-products
- Treat wastewater before release into the environment
- Monitor chemical use and by-product generation
- Implement responsible practices
Typically, responsible practices involve:
- Closed-loop water systems
- Safer bleaching agents
- Effluent treatment plants
- Regularly conducting tests on the environment
Clean production practices lower the probability of sudden stoppages or fines, which would affect supply.
Packaging reduction
Medical packaging must be designed to protect sterility, but this can also be wasteful.
Manufacturers are now working to redesign packaging to be more efficient. Lightweight cartons and optimal pouch sizes can greatly reduce shipping and waste.
For high-volume items, even small changes can result in significant overall reduction.
Some common methods for reducing packaging are:
- Using thinner, durable materials
- Standardizing carton sizes
- Using recyclable materials
- Using fewer secondary layers
Less packaging also means lower freight costs, which has a positive impact on the environment and the procurement budget.
Social Compliance Audits
Ethical Sourcing also includes the conditions of work in the factory.
For hospitals like ours and NGOs, we do not want the supply chain to be associated with unsafe work conditions or unfair labor practices. Thus, social compliance audits are the norm.
They may include:
- Working Hours
- Wages
- Safety procedures
- Child and Forced Labor
- Employee Training
For the production of sterile gauze pads, a stable and well-trained workforce is also a key factor in the consistency of the product.
Ethical Sourcing is therefore a key factor in the social responsibility and quality of the product at the same time.
NGO and Public Hospital ESG Requirements
Public tendering now includes ESG scoring in the tender requirements.
Environmental, Social, and Governance criteria are now part of the tendering process, alongside price and technical specifications.
NGOs and public institutions may request a sustainability report or audit results, or a carbon reduction plan, when buying sterile gauze pads.
This is particularly the case in Europe and international health programs. Suppliers who can demonstrate ESG initiatives may gain a competitive advantage.
Sustainability is gradually moving from a ‘nice to have’ feature to a qualification requirement.
Digitalization and the Future of Sterile Gauze Sourcing (2026–2030 Trends)
Healthcare procurement is also becoming more digital.
Manual spreadsheets and reactive ordering are being replaced by connected systems that offer real-time visibility into production, inventory, and demand. For frequently used items like sterile gauze pads, digitalization can help avoid stockouts and improve the accuracy of planning.
Digitalization is not just about the adoption of technology for the sake of technology. Digitalization reduces errors, speeds up reaction times, and increases traceability across borders.
As global sourcing becomes ever more complex, these attributes are rapidly becoming a source of competitive advantage.
The following are the trends that will influence the procurement of sterile gauze pads in the future.
Smart factories
Factories are increasingly incorporating sensors and connected equipment into daily operations. Smart production lines have automatic monitoring of temperature, humidity, equipment performance, and sterilization.
If a problem is detected, it sends a warning before a defect appears. In the case of sterile gauze pads, this ensures a consistent quality product and fewer rejects.
Smart production lines also enable management to have a real-time view of production. Smart factories eliminate uncertainty for both suppliers and buyers.
Automation and robotics
Automation is increasingly being used in cutting, folding, packaging, and palletizing. Robotics minimize manual handling, which reduces the risk of contamination.
This is particularly important when producing sterile gauze pads, where a cleanroom environment is required.
Typical benefits of automation and robotics:
- More production capacity
- Fewer human error-related issues
- Less labor-related variability
- Consistent product size
Though it is costly to install, automation is often associated with lower costs and stability.
Buyers often perceive suppliers who have automated production to have a stable supply.
Blockchain traceability
Traceability is increasingly detailed and transparent. Technologies such as blockchain enable the traceability of each batch of sterile gauze pads from the source of the cotton to delivery.
Data is stored electronically rather than being scattered across multiple records.
This improves:
- Recall management
- Compliance verification
- Anti-counterfeiting
Traceability for regulated healthcare markets facilitates audits and establishes trust between suppliers and buyers.
It also speeds up investigation time in case of quality concerns.
Digital procurement platforms
Digital procurement platforms are increasingly being used by procurement teams to manage procurements.
Digital procurement platforms offer a single point of access to supplier information, certifications, prices, and purchase histories.
For common items such as sterile gauze pads, they facilitate faster reordering and minimize paperwork.
Some of the features of these systems are:
- Automated approvals
- Supplier scorecards
- Electronic contracts
- Real-time inventory management
Digital systems minimize paperwork and improve accuracy.
Digital systems make it easier to compare suppliers objectively.
Predictive Demand Planning
Data analytics is helping buyers move from a reactive ordering approach to a more predictive planning approach. By analyzing past consumption rates, seasonality, and changes to the program, the software can forecast the demand for sterile gauze pads several months into the future.
This enables:
- Earlier production planning
- Fewer emergency shipments
- Lower inventory costs
- Reduced stockout situations
Predictive forecasting enables greater coordination between the supplier and the buyer. By working together with reliable information, the process is more streamlined and cost-effective. In the end, the buyer is able to use predictive planning to gain more control over the process.
Conclusion: Turning Procurement Strategy into Safer Patient Care
The process of choosing suppliers for sterile gauze pads is, in the end, a medical choice disguised as a procurement choice. The process of manufacturing, certifications, testing, and logistics planning all play a role in whether the items are received safe, sterile, and ready to use.
The difference between an average supplier and a reliable source is now clear. Reliability, clarity, and record-keeping are as important as price.
At BKAMED, we produce the best-quality sterile gauze pads with a rigorous quality and compliance process. We encourage hospitals, suppliers, and NGOs to contact us directly or ask for a quote for reliable bulk sourcing.

