Every year, millions of surgeries depend on one often overlooked product: the lap sponge. But for procurement teams, a wrong move in supplier selection can create a host of risks-inconsistent absorbency and linting, missing certification, shipment delays disrupting operating room schedules.
For buyers, many are left asking the same questions: Which manufacturers can actually scale? Who meets global regulatory standards? And how do you separate reliable producers from trading companies? This uncertainty is exactly what triggered this article.
In this guide, we will profile the Top 10 Lap Sponge Manufacturers in China. We closely examine the materials they deploy, the manufacturing process employed, the certifications they hold, and ability to support export demands globally. The goal is simple: to empower buyers to make confident, low-risk sourcing decisions.
Key Takeaways
- The top 10 lap sponge manufacturers in China include BKA MED, Winner Medical, Zhende Medical, Allmed Medical Products, among others.
- Main clinical uses of lap sponges include the absorption of large amounts of blood or fluids during surgical procedures, providing protection against tissue abrasion and temporary packing for hemostasis in abdominal or trauma procedures.
- Quality control standards emphasize fluid management through absorbency and linting tests for the safety of the fluids, tensile strength/ply integrity tests to check the durability under stress.
- Global compliance pathways include ISO 13485 for quality management, CE marking, FDA registration/510(k) for U.S. imports, UKCA for post-Brexit UK markets, and NGO-specific standards on ethical sourcing/traceability.
Top 10 Lap Sponge Manufacturers in China
This ranking evaluates the top lap sponge manufacturers in the Chinese market according to production levels, adherence to rules, and B2B ease. The big three excel in production volume, but specialized manufacturers may provide higher value to custom hospital requirements.
Each entry lists detailed descriptions of the product to facilitate B2B analysis: from woven floors to sterilization chambers.
1. BKAMED (Hubei Baikang Medical Instrument)

Hubei province is considered the heart of China’s medical textile industry. BKAMED, founded in 2010, bases itself on more than two decades of gauze weaving and processing experience localized in the area. That heritage shows when dealing with complicated processes for producing high-performance Lap Sponges. The facility works from Xiantao, the center for non-wovens and gauze.
It represents a completely vertically integrated model, managing in-house everything from the initial spinning and weaving of raw cotton to the final sterilization and packaging. This control keeps third-party contaminants out of Lap Sponge production. Often, hospitals request specific thread counts or unique loop colors for tracking. BKAMED responds well to these bespoke requests.
Their 850+ specialized employees find a balance between traditional craftsmanship and modern automation. The factory maintains a monthly capacity of 18 million pieces, supporting consistent bulk replenishment for large distributors. Precision is achieved by means of automated weaving looms. The catalog covers all diverse needs: from emergency trauma up to elective surgery.
Sizes range from a standard 22.5 x 22.5 cm up to abdominal pads that reach 45 x 70 cm. Options include 4, 6, 8, 12 or 16 ply sterile sponge or non-sterile bulk. They also have incorporated radiopaque threads and X-ray detectable chips for post-operative visibility. The ability to customize for custom ply and X-ray thread patterns exists.
The company is very good at deep OEM and private label support, providing custom thread counts such as 21s, 32s, or 40s, and custom labeling for hospital systems. The quality management is ISO 13485 certified, fully CE and FDA compliant. Sterilization validation encompasses Ethylene Oxide (ETO) and Steam methods for bioburden reduction.
BKAMED exports to over 70 countries, including strong sales to the EU countries, like Germany, France, Italy, the United States, Southeast Asia and even North America, as well as to NGOs for the urgent deployment of surgery sponge equipment.
Pros:
- Superior Customization(OEM/ ODM Service): BKAMED offers sponges meeting special requirements, such as varying loop lengths and concentrations of barium sulfate.
- Zero-MOQ Flexibility: The program allows distributors to introduce premium Lap Sponge with minimal inventory burdens.
- Advanced QC Traceability: Each batch can be traced back in terms of cotton lots utilized and bleaching cycles performed, making it easier to audit hospitals.
- Rapid Lead-Time Management: Through the logistics, there is a provision of 14-18 day fast track cycles in cases of bulk orders of
- Transparent Auditing: Digital portals provide buyers with online access to important production information in real time.
Cons:
- B2B Only: Lack of physical store outlets plus consumer awareness.
- Peak Demand: Capacity has booked up months ahead of time through Q4, so there’s a need to start planning early for Lap Sponge demand.
1. Winner Medical
Founded in 1991, Winner Medical is a multi-billion dollar conglomerate, employing more than 10,000 people. It has eight main production bases all over China, which come in hand particularly when hospitals require astronomical quantities of Lap Sponge.
The portfolio is an extension of the company’s patented PurCotton non-woven technology, which they lead in the traditional woven gauze sponges segment, where Lap Sponge is either pre-washed or non-washed, blue or white loops.
Different options for packaging include single-piece peel pouches or rigid blisters with five sponges each, increasing the efficiency of OR activities. With regard to capacity, one month can yield 55 million gauze pieces, fitting well with bigger hospital systems, while OEM is suitable for standard orders in high volume, except with rigid requirements in approval for special offerings such as Lap Sponge configurations.
The quality system carries certifications for ISO 13485, CE (MDR), FDA. The company holds the leading place for environmental certifications for carbon footprinting per Lap Sponge. They use ETO chamber-scale sterilizers and Gamma radiation for sterilization. The export market spans more than 110 countries, with strong business relationships existing for North America and Japan.
Pros:
- Market Dominance: Unmatched R&D makes innovation in types of lint-free surgical sponges.
- Patented Technology: PurCotton provides excellent lint-free options to replace conventional gauze.
- Colossal Infrastructure: Scale handles surges of global demand without breaking a sweat.
- Sustainability Leadership: Carbon-neutral certification across all its Lap Sponge production.
- Industrial Scale: Absorbs massive order spikes seamlessly.
Cons:
- Inertia in Corporations: Size slows changes to Lap Sponge specifications.
- The Winner Tax: Brand prestige hikes prices 15-20% over comparable Lap Sponge.
2. Zhende Medical
Established in 1994, Zhende Medical prides themselves on their reputation as the most European-centric manufacturer in China by focusing intently on the technical aspects dictated by the Medical Device Regulation in the European Union.
The primary production facility in Shaoxing, China, is a technical masterpiece of modern engineering. They employ a high-speed robotic system to process the folding and sewing of each individual Lap Sponge, achieving this degree of uniformity. Unlike other production units, Zhende produces their own weaving and bleaching to guarantee strict adherence to environmental and purity regulations.
The amount of items they can produce per month for a Lap Sponge is approximately 35 million objects. They can provide significant aid in private labels, especially to European distributor needs. They can, amongst other things, vary the ply, dimensions, or even the kind of x-ray detectable barium sulfate thread that is used within the weave.
Zhende were among the first Chinese manufacturers to make a full transition to the EU MDR regulations. The manufacturer is certified to the ISO 13485 standard and holds regulatory 510(k) clearances from the FDA. Their sterilization information is among the most extensive any manufacturer has to offer a business-to-business customer; ETO and Steam options are well-documented to meet customer needs.
Zhende is the main Chinese exporter to the U.K.’s National Health Services as well as many big health service providers in France and Spain. Their export team is highly professional and familiar with all paperwork requirements that are common in Europe.
Pros:
- MDR Compliance: Early Adoption is also consistent with current EU regulations for Lap Sponge.
- High End Automation: Robotics helps to reduce mistakes during folding processes.
- Surgical Kits: Bundles make procurement easier for OR setup procedures.
- Logistics Advantage: The advantage is related to the proximity to a port, which provides
- Automation Excellence: Uniformity of every sterile sponge fold.
Cons:
- Government Priorities: There is a possibility that delivery of a private Lap Sponge order will be delayed
- Siloed Communication: Account managers often delay in responding.
3. Allmed Medical Products
Perhaps the most efficient of all pure manufacturers in this ranking is Allmed Medical, who since 1997 has been among the first choice for Western brands that want to outsource a Lap Sponge manufacturing process under strict quality control.
Allmed has six huge production facilities, as well as more than 100 intelligent production lines. They describe themselves as a vertically integrated manufacturer, which, in this case, implies an ability to begin production of a Lap Sponge with raw, natural cotton fiber. They can control the bioburden of a Lap Sponge from the very first step of production. Their facilities include huge cleanrooms of 100,000 classes.
They provide an extensive range of woven gauze sponges. Lap Sponge products by the company are available in a bewildering variety of specifications such as 8-ply to 24-ply, essentially designed to meet the needs of specialized aspects of neuro surgery. They provide sterile and non-sterile variants, including bulk packaging aimed at meeting requirements of container loading/storage.
Their monthly output is around 30 million pieces. Allmed specializes in the white-label business model. They offer end-to-end support for the management of private labels. For example, they can manage the overall graphics for packaging and printing labels for you. Suppose you are interested in buying a Lap Sponge with particular barcodes or even RFID. Allmed can support you.
As a publicly traded company on the Shenzhen Stock Exchange, their quality systems are always under public scrutiny. They hold certifications such as ISO 13485, CE, and FDA. Sterilization validation of their products is world-class, exceeding even demand in their geographic region.
One of their strengths is that it is a major player in the North American marketplace, providing many of the biggest private-label brands used in American hospitals. It is also a player in both the Middle Eastern and African markets, where it is very cost-efficient.
Pros:
- Cost Efficiency: Has the ability to produce high-quality Lap Sponge at competitive prices
- Cleanroom Standards: Class 100,000 sets the gold standard for audits.
- White-Label Mastery: Manages all aspects of international branding/packaging.
- Vertical Control: Owns a spinning mill for Cotton.
- Private Label Expertise: Smooth transition from factory to branded Lap Sponge.
Cons:
- Accessibility: Preference is Tier-1 distributors; resists smaller buyers.
- Brand Awareness: Lack of product identification due to the nature of the contract.
4. Hubei Qianjiang Kingphar Medical Material
Kingphar Medical is based in Qianjiang, Hubei Province, at the very heart of the cotton-growing region. Since 2001, they have capitalized on that location to become the industry leader in raw gauze quality.
Kingphar’s production area is great, with upwards of 1,000 sets of looms and sewing machines. These are renowned manufacturers of Pure Oxygen Bleaching lines. In contrast to the conventional chlorine bleaching, this kind of processing enables a whiter, more absorbent Lap Sponge free from toxic residues. They manage the entire process in-house, thus enabling their zero-outsourcing policy for their core gauze products.
Kingphar has the potential of manufacturing approximately 20 million units of the Lap Sponge per month. They are extremely price-competitive, especially in the case of non-sterile bulk orders. Although the company does provide OEM services, its main strength is to provide high-quality products in the form of materials as well as finished goods at a price point which is hard to be matched by the urban factories.
It is also ISO 13485 and CE certified. Kingphar also has a long and successful clinical FDA compliance record themselves. Their test labs can detect everything from fiber linting to the chemistry level present in the water used to bleach the material. The ETO process is how they sterilize most of their sterile products.
They have a very strong foothold across South American, African, and East European regions. See, across these areas, it is observed that for these customers, the cost to quality for Lap Sponge is the primary differentiator, and across these places, Kingphar always wins on that front. They work for international aid organizations.
Pros:
- Bleaching Purity: The product obtained by the oxygen method has residue-free absorbent woven gau
- Aggressive Pricing: Best price per unit that can be offered on bulk non-sterile lap sponge
- Regional Expertise: Hubei sources of high-quality cotton purchased at low prices.
- Product Diversity Regardless of whether we’re talking about a general product line like S
- Value Pricing: Best at handling bulk purchase situations.
Cons:
- Technical Gap: There is a gap in digital order tracking
- Regulatory Support: Lagging for complex FDA 510k approval for Lap Sponge.
5. Yangzhou Super Union Medical Material
Yangzhou Super Union was formed in 2003. They developed their own identity by specializing in the exceptions of the rule. While others cater only to the most commonly found size, Yangzhou specializes in variety.
The company’s portfolio consists of over 300 items in various medical products, but among those products is its lap sponge collection that stands out in terms of size options offered. The company caters to the often-required smaller niche size needed in pediatric or orthopedic surgery cases.The surgical sponges types offered come in non-woven forms for those looking for solutions with reduced lint material in their facilities.
The facility has 20,000 square meters of floor area in Yangzhou. This is a mid-size operation, but it’s extremely agile. Where other large conglomerates won’t, Super Union will reuse their production lines to make smaller, specialized orders. They have a strict in-house system with regard to their own folding and sewing to guarantee that their hand finish quality on their Lap Sponge is always good.
Although their overall volume may be less than either Winner or BKAMED, they are the largest suppliers for Small-Batch Customization. They have the capability for custom labels and different packaging forms for a Lap Sponge order where the volume is less than a Tier-1 company can manage. They are the best fit for working with a medium-sized distributor.
They are certified by ISO 13485 and have CE and FDA registrations. Their quality control team is renowned for being very meticulous, even providing a pre-shipment sample on every order of bulk purchase of a sterile sponge. They mostly offer ETO sterilization validation.
Super Union has a substantial footprint in over 100 nations. They have managed to successfully operate in regions with differing regulations like Chile, UAE, and Malaysia. The fact that they understand different customs regulations makes them extremely easy to deal with in international trade.
Pros:
- Niche Flexibility: Runs smaller lines for specialized medical gauze.
- Responsive Service: Mid-sized service provides personal and direct communication.
- Quality Consistency: An audit clean sheet over two decades.
- Price Stability: Upholds its contracts even though prices fluctuate frequently involving raw
- High Engagement: Highly engaging for specialized requirements of B2B Lap Sponge.
Cons:
- Lower Total Capacity: May not compete with the giants’ level of production.
- Limited Logistics: The involvement of external agencies can cause delays in shipping.
6. Vench Medical Products
Vench Medical is a younger (established in 2012) and more ambitious player in the market. It has a state-of-the-art plant that focuses intently on automated packaging; they have invested in the cutting-edge technology in terms of heat sealing equipment, making it possible to package one Lap Sponge per minute, far beyond what conventional manufacturing capacity would facilitate.
They also offer a line of Lap Sponges, which are for the highest volume, standard specification requirements. These are 100 percent cotton, high absorbency, and are sewn in with X-ray detectable chips. They are a leading source for the sterile 45x45cm sized sponge, which they ship in huge quantities with short lead-time requirements.
Their capacity is an incredible 12 million per month. Of course, they are built fast, but they can also provide some decent OEM work. Their strongest skill is probably coming up with a high-graphic retail box or a box suitable for a hospital setting that houses a Lap Sponge that needs to present itself professionally.
They are completely certified to ISO 13485, CE(MDR), and FDA. Their validation for sterilization is state-of-the-art and very well documented. They often update their B2B customers in real-time regarding their batch’s status within their particular chamber.
Vench is highly competitive in the Middle Eastern and Southeast Asian markets. The ability to promptly deliver orders for a Lap Sponge product makes them favorites for regions with an expanding medical infrastructural base.
Pros:
- Production Speed: The latest models offer high production speeds in pieces per hour.
- Modern Branding – Professional presence. Simplifying international catalogs.
- Competitive in Mid-East: Tailored logistics have established themselves in this region.
- Cleanroom Integrity: Despite high-speed Lap Sponge manufacturing lines, cleanroom hygiene standards are maintained.
- Rapid Lead Times: Best lead times from production to ports in China*.
Cons:
- Standardization Focus: Resists complex manual Lap Sponge customizations.
- Market Concentration: Fewer Lanes for North American Shipments.
7. Jianerkang Medical
Established back in 1999, Jianerkang is one of the oldest and most respected private companies and manufacturers in the country. They describe themselves as the benchmark factory for Jiangsu, China.
Their footprint is enormous: a plant that processes at a 100,000-class purification facility extending over 80,000 square meters in size. They employ over 1,800 persons and have invested in high temperature and high pressure equipment in their bleaching plant, one of the best in the world.
JEK provides a vast range consisting of over 2,000 specifications, and the range of its Lap Sponge is extensive, covering varieties such as pre-washed, non-washed, sterile, and non-sterile. The OR Towel, which is oftentimes used together with the Lap Sponge by large hospital groups, is also manufactured by JEK. Its woven gauze sponges have a reputation for density and tensile strength.
As one of the largest OEM companies in China, their scale is enormous. They carry out customization for all aspects of the product, for instance, the kind of yarn used for the product, which can be between 21-40s, or the folding of the product to form the sterile sponge. They have experience dealing with the complexities involved with personalized orders for international brands meant for the private label.
They were one of the first companies to secure the prestigious ISO 9001 and 13485 requirements. In addition, they have CE and FDA registrations and numerous 510(k) and NDC numbers listed thereon. Further, they have qualified labs to conduct sophisticated analyses, including chemical and physical testing.
JEK is a player with a massive footprint in North America and also globally in Europe. They often provide goods as the primary supplier for Tier-1 distributors worldwide. Their longevity in the market allows them unparalleled insights into shifting global regulations.
Pros:
- Historical Reliability: Markets require three decades to go through every market cycle.
- Massive Assets: Top-class machines guarantee a consistent Lap Sponge.
- Strong Domestic Base: Scale lowers export prices.
- Wide Product Range: One-Stop Shopping for a Wide Assortment of Woven-G
- Proven Reliability: Consistent Performance for 30 Years
Cons:
- Domestic Focus: Chinese orders may precede B2B shipments.
- Bureaucracy: Time-consuming processes delay the opening of new accounts
8. Changzhou Health Microport Medical Device
While many of the businesses within this list are focused upon the textile aspect of the Lap Sponge, Changzhou Health Microport is specifically involved in the technology of the operating room.
Established as recently as 2008, they have a 12,000 square meter plant which is more reminiscent of a typical electronics plant than a textile mill, and they specialize in high-precision medical devices, bringing this mindset to bear on the creation of surgical consumables.
Their product, Lap Sponge, is designed specifically with today’s Smart OR in mind. Bioknowledge: The company is a RFID embedded leader in the field of medical sponges. Although a biologist would examine the sponging and lapping mouth parts of an insect to grasp its absorbing qualities, they use advanced sensors so that no surgery sponge is ever left behind in a patient again.
Significantly, their capacity is less in comparison with the other textile giants, since each of their Laps Sponge is considered high-tech. They specialize in high-value customizations, such as incorporating certain types of RFID chips that can be used with existing software used in hospitals. This is highly specialized work that cannot be expected in an ordinary gauze factory.
They also possess a certified manufacturing area according to ISO-Class 8 specifications and all relevant ISO and CE Certifications. Their quality systems are also consistent with devices used by the medical profession, ensuring that not only are the gauze materials dependable but also the tracking electronics provided with Lap Sponge products.
Their main customers are high-end medical centers located in North America and Western Europe, a place where the cost of a Retained Surgical Item legal case is well beyond the price of a high-tech sterile sponge.
Pros:
- Tech Innovation: Pioneers of Smart Lap Sponge category.
- Safety Focus: The sensors monitor in order to avoid retained items.
- High-End Manufacturing
- Custom Coating: Enhances surgical sponge efficacy.
- High Precision: One-stop solution for device-grade control on all products.
Cons:
- High Price Point: Tech adds premium prices.
- Limited Volume: Fewer units than Volume giants.
9. Ningbo Medelast Co.
Ningbo Medelast was established in 2002. They place a lot of emphasis on minimizing the impact of the bleaching and sterilization process on the environment. They use recycled systems and energy-saving ETO chambers, making them a favored supplier by many companies with Green Procurement programs.
They provide a high-quality standard lap sponge while emphasizing reducing linting and fibers. They have a clean portfolio, concentrating specifically on the most utilized forms of surgical sponges. They also provide biodegradable packaging alternatives, replacing plastic blister packaging with medical-grade paper if achievable.
The company also complies with MDR (CE) and FDA regulatory requirements. Their quality system is state-of-the-art. Their system is transparent, giving B2B buyers a digital look inside their sustainability metrics at times. They employ an ETO sterilization methodology that uses a superior aeration process to ensure zero residuals.
They are finding great success among the Nordic countries and Western Europe, where environmental regulations are the highest. They also work with many NGOs that pride themselves on ethically sourced and made medical materials.
Pros:
- Eco-Conscious: Ideal for hard ESG Hospital Targets
- Modern Facilities: Energy-efficient equipment operates the facilities.
- Market Agility: Eager for international B2B growth.
- Ethical Sourcing: Deep transparency in the Cotton Chain.
- Sustainability Leadership – Eco-Friendly Medical Textiles.
Cons:
- Brand Maturity: Continuously building global reputation.
- Scaling Pains: When Lap Sponge experiences fast growth, it can lead to laps
What is a Lap Sponge?

A Lap Sponge is a big format surgical absorbent that is normally used during invasive surgical procedures. By definition, Lap Sponge, as a medical supply product, is specially made to be usable within the body, specifically within open cavities of the body, where fluid management is important.
Unlike simple dressings, the Lap Sponge is constructed with specific standards for dimensions, absorbency capacity, and linting. The product is primarily constructed with cotton materials of medical grade and includes a radiopaque marker for detection during the surgical count.
Lap sponges can generally be grouped as operating room consumables rather than general wound care products. As moving parts subject to operating requirements, they thus command greater levels of compliance requirements, quality testing, as well as degree levels of batch tracing.
In practice, these products tend to be provided in bulk form, generally for institutional sales, typically in a sterile form as a lap sponge or in a non-sterile form depending upon downstream protocols.
How Lap Sponges Differ from Gauze Sponges

Since their sources of raw material are similar, lap sponges and gauze sponges have very different uses in medicine.
A simple and often used reference point can be illustrated as follows:
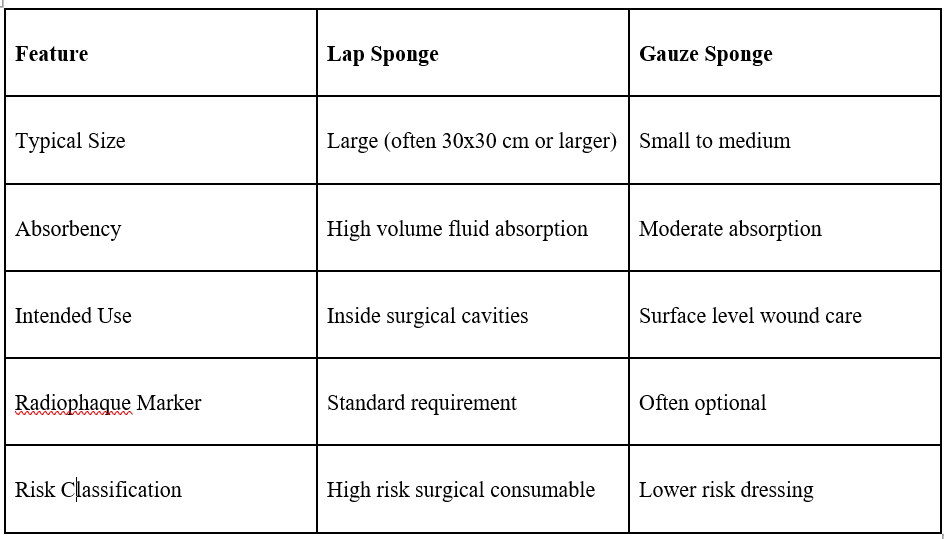
Lap Sponges are specifically designed for deep surgical areas, which are usually heavy in terms of fluid loads and also present obscuring visibility conditions.
Gauze Sponges, including Woven Gauze Sponges, are more appropriately used for external cleaning, dressing, or procedural support.
The rationale for this differentiation lies in the way in which lap sponges have been defined with reference to other surgical sponge types that need special care while handling or administering by different authorities in respective surgical institutions around the world.
Core Clinical Applications
The design of a Lap Sponge is a reflection of its use in the operating room. Each application requires predictable performance under pressure, literal and figurative.
● Blood and Fluids Absorption
Some of the uses of lap sponges during open surgeries are to act as the first line tool in managing blood and fluid accumulation. Their larger surface area, multi-ply construction allows for rapid absorption, thus not needing frequent replacements.
This reduces clutter in the surgical field and supports better visibility for surgeons and assisting staff. In high-volume procedures, consistent absorbency is not a preference-it’s a necessity tied directly to procedural efficiency.
● Tissue Protection During Surgery
They are also employed as physical barriers or sponges. Most of them are placed between instruments and delicate tissues in order to prevent abrasion, drying, or accidental trauma.
In this function, the sponge is a passive protector. Its softness, its lint control, and moisture retention must remain consistent in the process even when repositioned multiple times.
● Temporary Packing and Hemostasis
In certain surgical procedures, there can also be the temporary use of a Lap Sponge to control bleeding or stabilize involved areas, particularly in unintended areas such as in abdominal or trauma surgeries, where closure is not immediately possible.
The thing is, this particular application puts the sponge inside the body cavity entirely. Thus, radiopaque visibility as well as surgical counting are more significant. The slightest change or variation can spell ruin.
Why Lap Sponges Are Classified as High-Risk Consumables
From a governmental or procurement basis, lap sponges are classified within a higher-risk group than many other types of disposable products that may be used during a surgical procedure. This classification is based on two main criteria: counting practices for surgery, or risks associated with retained items.
● Surgical Counts
Each surgical count is performed according to formal procedure, where all items inserted into the surgical site are counted. Lap sponges always have to be included because they would be inserted into the body.
Each Lap Sponge should be able to be identified, counted, and located easily. This explains why radiopaque threads are mandatory, along with thorough examination of consistency in dimensions between batches.
However, now when these counts do not reconcile, the consequences do not stop in the operating room. They go into delay, imaging, and incident reporting, which include costs and compliance issues.
● Retained Surgical Item (RSI) Risks
Retained surgical items, one of the most critical surgical errors, can cause problems, especially in cases like a surgical sponge left inside a patient, which can lead to potential harm, litigation, and damage to reputation.
The question of lap sponge usage in RSI’s prevention strategies arises significantly with their size and placement in the body. The procurement team in this case assesses different sources not only in relation to cost but also in relation to their performance as a marker, material sources, and their ability to maintain consistency in quality in the long term.
See, this is the point at which supplier selection becomes a risk management decision, as opposed to a sourcing decision. Suppliers who do not demonstrate process control and auditing capability expose manufacturers to downstream clinical and regulatory problems.
Materials Used in Lap Sponge Manufacturing
The performance of a Lap Sponge commences long before it enters a working environment because, for a B2B buyer, understanding what goes into a Lap Sponge is, for them, far from being a task based on mere expertise but rather a means to evaluate the level of compliance within a given regulatory framework.
Part of this section will help break down and describe the overall materials involved with the creation of Lap Sponge and how this can affect the absorbency and detection rate around the world.
Medical Grade Cotton

Medical cotton still forms the basis of most forms of lap sponges. It is easily absorbed and is soft on tissues; therefore, it comes into contact with tissues internally as a matter of course.
However, not all cotton is the same. The performance of a Lap Sponge depends in great measure on the selection, handling, and control of the cotton.
● Fiber Length And Absorbency
Fiber length is one of the most important but least noticed characteristics of cotton fibers used in selection. Longer fibers have stronger interlock properties during weaving, producing a fabric with high absorbence of fluids together with strength.
In practical terms, longer fibers ensure a Lap Sponge is capable of retaining larger volumes of blood and fluids without deteriorating or disintegrating and leaking lint. This is particularly useful to ensure a lap sponge is not subject to any undue handling or maintained over long periods of time.
Shorter fibers, by contrast, may affect absorbency and linting properties. For the purchasing executive, the consequence is variability of clinical performance, which makes manufacturers insist on tight control of the fibers used for manufacturing.
● Whiteness & Impurity Control
Being white does not relate in any way to aesthetics. What whiteness index provides is a substitute, a symbol of being clean and pure. Cotton of a medical grade, which is used in a Lap Sponge, follows a washing process as well as a chemical bleaching process in order to rid the substance of any seeds, plant materials, and oils found
Impurities remaining in the cotton can affect absorbency patterns as well as introduce foreign materials into the operating site. As such, cotton is tested before it is woven for ash, foreign fibers, or chemicals present in it.
Furthermore, in regulated markets, purity requirements are also linked to biocompatibility properties, particularly if it is intended as a sterile sponge directly applied in an internal capacity.
Non-Woven and Blended Options
Although woven cotton fabrics have traditionally remained the standard, some fabric manufacturers often explore different fabrics, especially non-woven or woven fabrics, which can offer certain functions by joining their own cotton materials with man-made fibers.
However, non-woven materials are less common for lap sponges compared with surface dressings. This is due to the fact that woven materials offer more dimensional stability.
For the buyers, the only concern will be the consistency of the materials, and any other variation from the traditional form of cotton will require the availability of validation, clinical, and regulatory data to avoid the risk of a new innovation in materials, which will make the purchased materials a risk to the entire supply chain.
In certain products, non-woven layers are selectively used, but the major part of the layers that act as an absorbent stays true to its cotton nature.
Radiopaque Threads and Markers
The incorporation of radiopaque parts is not cosmetic for lap sponge manufacturing. They are safety features designed for detection during and after procedures.
The lack of reliable radiopacity makes a Lap Sponge fail to live up to generally accepted expectations for surgical risk management.
● Detection Requirements
Radiopaque threads or markers are designed to be X-ray detectable. If there are questions regarding a surgery’s counts, imaging serves as a last line of defense to detect retained items.
In order for detection to be effective, the location of the radiopaque element must be consistent, secure, and unbreakable. If this is compromised, detection becomes a problem.
Threads intended for visibility, known as radiopaque threads, are typically integrated directly within the sponge structure, not surface-attached.
● Compliance Expectations in Surgery
From the viewpoint of compliance, the ROP properties are intrinsically linked with the surgical safety standards used by the surgical team. Both hospitals and NGOs mandate international standards for ROP properties for the destination countries.
The regulatory authorities do not specifically test for radiopacity on its own. The product validation process includes its material compatibility, durability, and labelling accuracy, etc.
Non-radiopaque failures aren’t just quality issues in the ordinary sense. They’re also reportable failures in a range of markets, potentially triggering audit trails, product recall, and damaging buyer confidence.
Material Traceability and Batch Control
Material traceability is where manufacturing discipline manifests itself: For a Lap Sponge, this means traceability between raw materials, e.g., cotton, and/or incorporated radiopaque materials, process variables, and production lot number.
The relationship also allows manufacturers and users to take immediate action in case quality issues are raised. Additionally, it facilitates regulatory documentation, such as ISO 13485 and export-focused compliance schemes.
Which of the following would be an effective element of
- Qualification of cotton and other material suppliers
- Incoming material inspection records
- Lot-level identification during production.
- Description of samples retained by the provider of a service
With respect to global supply chains, traceability can aid recalls should they ever be required. Without traceability, a small problem can grow into a major problem of dislocations.
Now, this is where experienced buyers draw a line of demarcation between commodity suppliers and long-term partners. Suppliers who have made an investment in traceability systems have demonstrated an understanding of surgical risk, as well as production efficiency.
The Lap Sponge Manufacturing Process
Behind every Lap Sponge is a manufacturing process that is tightly controlled, aimed at delivering consistency, safety, and compliance on a large scale. To B2B buyers, knowledge of the manufacturing process can aid in differentiating qualified manufacturers by those that either use minimal controls or not at all.
Within this section, each stage of production is given an explanation with regard to what exactly it entails in relation to lap sponge manufacture, ready for distribution around the world.
Selection and Pre-Treatment of Raw Cotton
The process starts by choosing raw cotton material. Medicals-only raw cotton material was used to ensure its purity and length meet the benchmark requirements to manufacture a lap sponge.
Before any mechanical processing takes place, pre-treatment of the cotton is necessary to remove any naturally occurring impurities like waxes, seeds, and plant material. All of this needs to be removed to ensure that a quality base for the surgeons’ material remains.
At this point, manufacturers also conduct an incoming inspection. This step ensures the consistency of the length of the fiber and the moisture content, which in turn plays a crucial role in the following processes of making a Lap Sponge.
Carding, Weaving, and Ply Formation

Once they have been pre-treated, they enter carding, in which they line up in neat webs, untangling them, so they will uniformly absorb and/or hold up well in a straightforward sense.
The carded cotton is then woven into gauze fabric. It is important to note that in most situations, a controlled weaving process is employed; it is similar to an ordinary woven gauze sponge but is more precise, with higher weight per square meter.
The layers are then combined in multiple layers to produce the desired ply structure, which is required by a Lap Sponge. The formation of the ply structure of a Sponge is significant, reflecting its absorbency and strength, depending on the number of plies present.
Cutting, Folding, and Edge Control
This woven, layered fabric is then cut into standardized dimensions. Here, precision is important since any variation in dimensions can affect surgical counts.
After the fabric is cut, it is folded to its final form. The patterns used in folding are such that the fabric has the largest possible surface area.
This is where edge control is taken into consideration by the manufacturers, where designers ensure there is control either through folding stitches to minimize fraying and lint fall-off. Lack of control can translate to lint fall-off, and this is never acceptable, especially considering its application as a surgical material.
This is the step in which the Lap Sponge starts to develop its ultimate recognizable form.
Radiopaque Thread Integration
Radiopaque thread integration is a dedicated and closely monitored stage of the process. The incorporated radiopaque element enables the Lap Sponge to be detectable via X-ray if the surgical counts remain indecisive.
Threads are generally integrated into the sponge construction, not glued into place after the fact. This prevents detachment and provides a consistent appearance throughout the life of the product.
For that, thread placing, tensile strength, and imaging visibility are tested by manufacturers during and after integration. Any inconsistency in this stage compromises the reliability of detection and introduces serious compliance risk.
See, radiopaque integration is not a value-added feature; it’s a non-negotiable safety requirement for lap sponges used in invasive procedures.
Washing, Bleaching, and Drying
After manufacture, lap sponges are subject to several washes. These washes eliminate the residual processing agents, loose fibers, and microscopic impurities introduced from previous steps.
Bleaching is subsequently applied, using controlled chemical processes that result in the desired required whiteness level without loss of fiber structure. Excessive bleaching weakens fibers; insufficient bleaching can let impurities remain in the fiber.
This phase is completed by drying. The processes of drying under control ensure that the sponge retains its shape, ply integrity, and absorbency. The moisture content is cautiously processed as the residual moisture might affect sterility and shelf life.
At the end of this phase, the Lap Sponge is clean, stable, and ready for sterilization or further processing.
Sterilization Processes
Sterilization is also applied in the case of lap sponges if they are being supplied as sterile products. The choice of sterilization techniques is dependent on market demand, compatibility, and buyer requirements.
● Ethylene Oxide (ETO)
Ethylene oxide can be used to sterilize items that are not tolerant to heat and humidity. ETO diffuses deep into the porous structure of the sponge so can be used on multi-ply items.
The used lap sponges then undergo a process of aeration to remove any gas left behind. The process should be validated by manufacturers because ETO leaves are heavily regulated around the world.
High-volume export processing may also employ ETO as the method provides consistent sterilities without affecting the materials.
● Steam Sterilization
The method in which steam sterilization uses high temperature saturated steam for reduction of microorganisms consists of using certain material capable of withstanding such processes.
Some types of lap sponge are available in formats used with steam, notably those supplied to institutional users who have autoclave procedures in place. Steam has an impact on the softness and overall absorbency of fibers.
manufacturers steam cycle validation ensures that products are sterile but that no damage occurs to their sponge structure.
Packaging and Labeling of Bulk Exports
Finally, the packaging step marks the end of the product development process before distribution begins, which is an important step to safeguard the product.
Usually, lap sponges come in inner packs or cartons that are used in bulk procurements. Materials used in packing aim at keeping them clean, preventing compression during packing, etc.
Labeling consists of specifications, batch numbers, the method used to sterilize the product, and any marks to show compliance. Proper labeling is important in the customs clearance, storage, and hospital records.
Packaging in export scenarios also has to withstand long-distance logistics without reducing quality in any way. A well-packaged Lap Sponge is always ready to use, not inspect or recreate.
Quality Control and Performance Standards
For B2B customers, quality control represents when vendors’ assertions are validated or repudiated. In terms of a Lap Sponge, underperformance risks are more tangible because they can be translated immediately into risk in terms of providing care, risk of non-compliance, or risk in terms of how that impacts service provision.
The following section will briefly explain how quality and performance in lap sponge production are measured, with specific reference to what tests and controls matter most to hospitals, NGOs, and surgical distributors.
Absorbency Tests and Linting
Absorbency is the most obvious characteristic of the Lap Sponge, yet it won’t be assumed for the purpose of product development. It will be tested by determining the rate of fluid absorption for the product.
The test replicates a surgery environment where blood, fluids, and fluid management are essential for a successful surgery outcome. A superior-performing lap sponge is one that has high absorbency rates, yet it does not collapse nor lose fluids it has absorbed into the field of surgery.
Linting tests are also run parallel to absorbency checks. Lint release inside the body cavity is unacceptable because loose fibers can interfere with healing or initiate inflammatory responses. Where the sponges will be used next to sensitive tissue or organs, it is very important that the linting performance be controlled.
For purposes of sourcing, inconsistent absorbency and/or excessive linting are usually indicative of poor fiber control or inadequate process discipline earlier in the production chain.
Tensile Strength and Ply Integrity
Tensile strength defines how well the lap sponge will hold together when pulled or twisted or needs to be repositioned during surgery. These are rather common real-world stresses of surgical procedures, even if they are not always immediately visible to the procurement teams.
Integrity testing of ply ensures that layers do not separate under stress. A weakly bonded Lap Sponge may tear or delaminate, which could increase the risk of fiber retention in the surgical site. Dry and wet tensile tests are performed because the material behaves differently when saturated. Strength that disappears when wet is a warning for institutional buyers.
Now, while this may sound similar with other lower-risk items, lap sponges are quite different in the sense that a deficiency in tensile strength goes far beyond being just a flaw in quality but may even slow down surgical processes while jeopardizing the patient’s safety.
Sterility Assurance Levels (SAL)
In cases where lap sponges are provided in sterile form, sterility assurance level plays an important role. Indeed, SAL quantifies the probability that products are nonsterile after being sterilized.
It is expected in most controlled markets that a Lap Sponge complies with a SAL of 10⁻⁶. This implies an expectation of less than a one-in-a-million possibility of any viable microorganism being present in the sterilization process.
Validations to ensure SAL include biological indicators, process challenge devices, and monitoring. All this information is well-documented and reviewed through auditing carried out by either the buyer or regulatory authorities.
If the product, like the Lap Sponge, is given a label of ‘sterile’ but does not support this claim through the validation of the SAL data, then the risk is
Bioburden and Microbial Testing
Before sterilization itself begins, a process known as bioburden assessment takes place. This is a measure of how many microbes are actually on a particular product. Products with less bioburden make for more reliable sterilization.
Bioburden testing helps manufacturers understand how clean they need to keep their upstream process. Anyone seeing a high number likely has problems with how they handle, wash, or control environments.
Post-sterilization microbial tests prove whether the process of sterilization was successful. These tests become necessary when exporting products since there is a need to validate each sterility claim.
Considering overall surgical instruments, instruments, and wipe materials, lap sponges are one of the exclusive materials that have significant internal usage and higher volumes of usage. Thus, the limits are so stringent.
In-Process vs Final Inspection Protocols
Quality control is not an event that happens after manufacturing. Quality manufacturers control quality throughout the manufacturing stage.
In-process inspections include monitoring critical processes such as weaving consistency, marker band placement, folders, and cleanness before sterilization, which prevents a defect from being made worse.
The final inspection involves properties of a finished product. Based on this property, one expects a verified size in addition to packaged properties with no inconsistencies in labeling or batches. The defined specifications need to be satisfied by all other attributes within a Lap Sponge that undergoes a final inspection with no areas for compromise expected from the inspection process
The problem is this: a final inspection-only strategy is a weak basis on which to build a quality program. In other words, if the defective material was not detected on its way through a process, perhaps thousands would already have been made by the time it is uncovered.
Global Compliance, Certifications, and Regulatory Pathways
For institutional buyers, compliance is more than just a check-the-box requirement at the end of a sourcing cycle. For these buyers, it is the guide to ensure that a Lap Sponge can enter a market legally, gain acceptance at hospitals across those markets, and pass audits smoothly. Verification of regulatory alignment can provide assurances to those buyers on how seriously a manufacturer takes risks, documentation, and long-term supply chain obligations.
Sub-section detailing key global certifications and pathways in lap sponge manufacturing and trade that need to be understood by B2B business decision-makers in simple language.
ISO 13485 & Quality Management Systems
ISO 13485 is the basis of medical device manufacturing compliance. ISO 13485 describes how a manufacturer handles quality, including device design, manufacturing, testing, and after-market steps.
For a Lap Sponge, ISO 13485 certification would prove to a potential customer that a quality management system is in place where there are controls, procedures, and assignable actions. Additionally, it would prove that processes are repeatable, operator-independent.
ISO 13485 specifications are usually considered as an essential prerequisite that has to be met, and buyers commonly accept them as such. Without it, it would be hard to on-board, audits would be protracted, and there would be little certainty about
More importantly, ISO 13485 connects production processes with risk management. The reason for this connection becomes particularly important with lap sponges, in that their failure has direct surgical implications.
CE Marking for the European Market
The CE marking will also enable the product, Lap Sponge, to enter the European Economic Area. The CE marking will also ensure that the product meets safety and performance standards and risk management principles as required by the European Union.
To obtain CE marking, manufacturers need to ensure that they classify a lap sponge, compile technical documentation, test it, and justify its testing. The assessment is done by examining its own claims of being radiopaque, safe, and sterile. CE marking is of paramount importance for European buyers, as without it, procurements cannot be made through formal hospital channels.
In the context of sourcing, it is also a matter of documentation discipline. In fact, companies having good technical records usually perform better in customer audits and tenders.
FDA Registration and 510(k) Considerations
To access the USA, FDA registration is required. The facility and product need to be registered with the FDA, and the company has to comply with Quality System Registration requirements.
In many situations, these types of lap sponges will be classified under medical device categories that do not actually require premarket approval but do require controls. Even so, A Lap Sponge may require submission under section 510k, which is intended to show substantial equivalence to an existing legally marketed device.
FDA requirements also include an insistence on traceability, complaint management, and process validation. These requirements are beyond manufacturing scope. For buyers looking at U.S. healthcare systems in particular, FDA registration may be seen as an equivalent of manufacturing sophistication even when purchasing in other areas.
UKCA and Post-Brexit Requirements
Following Brexit, it was observed that the United Kingdom adopted the UKCA marking as its own marking convention. This marking convention has different administrative and documentation requirements compared to CE marking.
A Lap Sponge delivered to the UK market has to abide by UK’s labeling requirements and conformity requirements even if it is displaying the CE mark. There is an overlap in requirements in the transition period, although it is expected now for it to conform to UKCA mark requirements.
Companies that service both EU and UK markets will often have to contend with parallel documentation models. In some cases, they may not be well equipped to handle it. Clarity on current UKCA marking status is also important for distributors and purchasing teams, since it can prevent import and customs issues.
NGO and Humanitarian Procurement Standards
The NGOs and humanitarian organizations have their own procurement standards, mostly beyond the basic compliance with regulations. These standards focus on safety, reliability, and ethical manufacturing.
Examples of NGO requirements for a Lap Sponge may include, but are not limited to, the validation of sterilization, batch traceability, and assured supply capacity. All documentation should be legible and easily available since audits can be virtual or performed within tight schedules.
Some of them also demand proof of ethical sourcing and labor practices. Even though these may not be made binding by the regulators themselves, such expectations arise within humanitarian tenders when selecting suppliers.
In this regard, lap sponges are handled as critical surgical consumables and not general supplies, thus raising the bar for alertness and time for consideration.
Documentation Required for Importation
Documentation is the medium by which compliance becomes portable. Without good paperwork in hand, an otherwise certified Lap Sponge might have to face customs hurdles.
Typical import documentation can include:
- Certificates of Conformity and Compliance
- ISO 13485 Certificates
- CE OR UKCA Declarations
- FDA registration details, if any
- Sterilization Validation summary
- Batch and lot identification records
The documents facilitate customs clearance, warehouse intake, and hospital inventory management. Inconsistencies or lack of updated documents may cause delays and affect landed costs.
The thing is that buyers may often find out about gaps in documents too late, that’s when they have already made their order!
Packaging, Labeling, and Logistics for International Buyers
As an institutional buyer, packaging and logistics are real issues, not an afterthought in design or operation, since they affect product integrity, government regulatory acceptability, or final cost of ownership. With Lap Sponge, package appearance is actually synonymous with ready-to-use or requires significant rework.
The following section is about how the packaging and logistics operations can be carried out concerning international lap sponge supply chain management.
Inner and Outer Packaging Formats
Packaging styles for lap sponges normally include various styles of multi-layer packages intended for retention of clean and organized aspects. Inner packages normally hold a specified quantity of sponges in a single package, while outer packages aid in their bulk moving and storage.
Inner packs, on the other hand, aim to prevent compression and contamination. In addition, materials used for packing sterile products must remain sterile until used. Packaging seal and material compatibility is also keenly monitored.
The outer packaging design emphasizes durability as well as efficiency. The cartons are of dimensions appropriate for palletization and containerized cargo, preserving Lap Sponge from moisture, pressure, or deformation during distribution.
Labeling Requirements by Region
Labeling is where you see regulatory compliance. The information sent with every Lap Sponge shipment must be correct and relevant for customs and internal audits.
These may vary from manufacturer to manufacturer but commonly include information such as the name and size of the item, size in ply units, method of sterilization, batch or lot numbers, and information regarding the manufacturer.
Regional differences are also important, as different regions have different requirements: in Europe, there are CE markings; in the UK, there should be alignment to UKCA; in America, there are FDA registration details and traceability markings.
One of the most commonly occurring causes for a delay in the shipment is mislabeling. It should be noted that if a product meets all requirements, it may be mislabeled during its shipment.
Shelf Life and Storage Conditions
Shelf life states the period for which the Lap Sponge is valid without affecting performance and safety. It is established through stability testing, and it is also related to the quality of the packages used and the method of sterilization.
While sterile lap sponges usually have an expiration date, non-sterile items can be managed in terms of provisions made in terms of degree of storage. Factors that control stability include temperature, humidity, and light.
Generally, instructions on storage requirements to ensure maximum absorbency and material integrity are specified on the packaging labels. The buyer has to take necessary steps to ensure maximum maintenance of such requirements from the transporters and the
Clear top-and-bottom labeling helps in stock rotation, reducing wastage, particularly where there are bulk procurements serving many sites.
Container Loading and Shipping Optimization
Efficient loading of the containers also decreases the freight costs by reducing the potential damage. Manufacturers calculate the size of the carton in order to optimize space utilization. For instance, in the case of a lap sponge, an over-compression shipping effect can influence its thickness as well as its usage. This is why loading is often well defined.
In addition to this, transit time is a factor taken into account by optimization as well. With long routes, there would be exposure to environmental changes, and thus a package would need to be
That being said, it appears that customers also do not understand how important shipping efficiency impacts price per unit. In fact, shipping efficiency can even offset rising compliance or material costs.
Managing Long-Distance Supply Chains
Global or world supply chains are complex. For example, the journey of a product like Lap Sponge can include several ports and check points before reaching the end user.
Those manufacturers who are more geared towards supporting long-distance supply chain operations provide documentation, communication, and lead times. This may help reduce uncertainty in hospitals and NGOs.
As far as lap sponges in general, companies with a history of successful logistical support will provide performance with consistency, even in the most turbulent of times.
Thus, a good logistics management system transforms a potential vulnerability into a tool for controlling risks.
Pricing Structures and Cost Drivers in Lap Sponge Procurement
The price of a lap sponge is influenced by a lot more than its unit cost. To the buyer in a B2B transaction, however, it also helps them understand the basic drivers of cost underlying price variation and aids better sourcing decisions.
This section breaks down how Lap Sponge pricing is structured and the dynamics that will influence total procurement cost.
Fluctuation in raw material costs
Medical-grade cotton is the main raw material in most lap sponges, whose prices continue to fluctuate depending on global supply, changes in weather conditions, and the demand cycle. Higher cotton prices lead to a squeeze on margins for manufacturers. Those with diversified sourcing or long-term supplier contracts are better positioned to manage volatility.
Sudden changes often just reflect upstream material shifts rather than opportunistic pricing for buyers. Transparency around raw material sourcing helps build trust in long-term agreements.
Sterilization and Compliance costs
There is an additional quantifiable cost associated with sterilization. For example, equipment such as an ETO chamber or steam sterilization equipment is validated, operators must be trained, and monitoring is carried out. Compliance costs go beyond sterilization. Compliance costs include certification, technical documentation, and auditing, which factor into the final cost of a Lap Sponge.
Manufacturers that reach many different markets face a higher overhead but reduce risks to buyers seeking to conduct business across regions.
MOQ, Volume Discounts, and Contract Pricing
Minimum quantities ordered have an impact on pricing levels. Larger quantities permit the reduction of production costs to a minimum.
Volume-based discounts are common in long-term contracts, especially where demand is forecasted by the contracting party to the business. Contract pricing helps to avoid short-term shifts in the marketplace.
However, unrealistic MOQs may indicate limited production capacity or poor quality management for lower-priced offers.
Total Cost of Ownership (TCO) for Buyers
The price per unit does not give us a complete picture, while total ownership costs involve issues such as logistics, storage, and risk exposure.
A Lap Sponge with a price increase while maintaining quality control and documentation may alleviate any hidden cost factors due to delays, audits, or rejection. The trend is towards evaluating suppliers on total cost of ownership as opposed to price alone: a practice which mirrors business decisions on equipment reliability.
In the high-risk conditions present in surgical settings, the ability to perform with dependability, coupled with the importance of continued supply, may even
Common Risks When Sourcing Lap Sponges from China
China is still an important manufacturing center for medical consumable products, including for lap sponges. It is indeed large, has cost advantages, and has export know-how. However, purchasing from this market also brings certain types of risk that B2B buyers ought to know about.
The key issues that are likely encountered whilst sourcing a product like Lap Sponge from China include:
Inconsistent quality in batches
One of the more commonly occurring issues relates to batch inconsistency. A good-performing Lap Sponge in one shipment may underperform or be affected by issues related to absorbency, linting, or size in another.
Such discrepancies are sometimes the result of inadequate control over the production process. For instance, changes in the cotton used might have an impact on its final performance.
For such players as hospitals or NGOs, batch variability induces friction in their operation. There is loss of trust in the product by their professionals, as well as pressures put upon in-house procurers in choosing suppliers.
The problem is this: companies with good quality processes document all manufacturing variables, while companies with weak quality processes depend upon end-of-the-line checks, which aren’t good indicators of fundamental processes.
Certification Gaps and Misrepresentation
Another type of associated risk is certification information that does not actually pass any level of verification or proof. There are some instances where certain suppliers tend to hand in outdated or borrowed certificates in any early stages of negotiation.
What does this mean? On one hand, it means that the company’s Lap Sponge, in its documentation, will appear to comply while in actual fact failing during scrutiny by purchasing companies or during regulatory audits. The most common lapses can be in terms of lapsed ISO certifications, in terms of scope, or in terms of having certifications but in other
Misrepresentation is not always intentional because there may be a misunderstanding from the supplier themselves about the scope and breadth of each supplier’s own certifications. However, whether it is done intentionally or not, the end result is the same.
Issues that are discovered in the late stages of a transaction can lead to delayed, rejected, or even recalled orders, apart from costs, especially in the case of institutional investors, where reputation is also a risk factor.
Deficient Performance of Radiopaque Markers
Performance regarding radiopacity is an absolute necessity for surgical sponges used for an invasive surgical procedure. Again, here is an industry where quality compromises happen.
Lackluster markers may be associated with their inconsistent placement, attachment, or visibility during imaging. Occasionally, markers are known to be pulled off during usage or are invisible on an X-ray. The lack of dependability of radiopacity of the Lap Sponge, which can compromise surgical count practice and patient safety, transforms a product sourcing problem into one of safety.
Those buyers who have not asked about validation data or imaging test results only learn about those problems after clinical application; from there, it is much more difficult to institute corrective actions.
Logistics Delays and Documentation Errors
Logistically even when the quality of a product is acceptable, it may still introduce risk. The main cause of shipment delays in sourcing a Lap Sponge from China is due to documentation errors.
Custom inspections can be triggered by something as small as a missing batch number or inconsistent labeling, or incomplete compliance paperwork. These delays disrupt supply schedules and increase landed costs.
Long transit routes also expose shipments to handling stress and environmental variation. Product integrity may be compromised before arrival without robust packaging and clear logistics coordination.
Today, mature exporters alleviate these risks through standardisation of documentation, checks on shipment in advance and clear communication with buyers. Less mature suppliers often can only react after problems occur.
Why BKA MED Is the Gold Standard for Lap Sponge Manufacturing
In a market crowded with suppliers, very few manufacturers demonstrate the operational discipline required for high-risk surgical consumables. BKA MED is special in its approach to manufacturing Lap Sponge products that reflect the way hospitals, NGOs, and distributors work in real life.

End-to-End Manufacturing Control
BKA MED controls the complete process of manufacturing lap sponges. From the raw materials selected, each step is undertaken within a highly regulated production scenario.
This measure of control minimizes the degree of variability. Procedures govern the cotton sourcing, weaving, folding, incorporation of radiopacities, as well as the sterilization and packaging.
For example, for producing a Lap Sponge, appreciable considerations would be those that can affect process change. Since BKA has control over making things, it limits factors outside that tend to introduce risk.
With reference to procurement, having end-to-end control simplifies audits and expedites qualification.
Consistent Quality in High Volumes.
The area in which many manufacturers have struggled with their processes involves scaling their facilities while maintaining the level of quality desired. The systems at BKA MED have been designed with the need for quality in mind.
Lap Sponge material preparation for each order occurs within standardized parameters with in-process inspection verification to guarantee that product performance levels do not change between small pilot orders and large institutional orders. If there are many hospitals to deal with, then similarity between shipments is vital. Otherwise, there is disretraining, disvalidation, and escalation.
The focus of BKA MED on repeatability ensures that buyers can plan over the long term by means of consistent supply performance.
Strong Compliance for Global Markets
BKA MED integrates regulatory readiness into their model for their manufacturing. Here, regulatory compliance is seen as an operations feature as opposed to an additional requirement for the market.
There are complete documentation facilities provided in Lap Sponge, made by BKA MED, in accordance with global regulations. These facilities include a quality management system, sterilization validation, and traceability records.
Since the compliance is standardized, the ability to move the product across regions is facilitated with minimal work involved, which is particularly useful for distributors wanting to handle different countries’ portfolios.
Now, a stronger regulator protects buyers too. When a company is audited, documentation can be provided, it is accurate, and it matches actual production processes.
Customization for Hospitals, NGOs, and Distributors
Although consistency is vital, flexibility is equally important. Customization is an option, but it is done in a manner that does not compromise main quality controls. Options that can be included for customization for Lap Sponge products could be specifications for sizes, ply, radiopaque markers, etc., and these can be included within validated processes.
In respect of NGOs or hospital groups, customization can represent protocols or perhaps storage considerations, and in respect of distributors, market differentiation or perhaps legislative alignment.
The key difference is that customization does not create uncertainty; it revolves around change control processes, i.e., the approvals provided by the buyer.
Long-Term Supply Reliability
The reliability of supply is subject to test when things get disrupted rather than being stable. BKA MED has developed capacity planning strategies, material sourcing strategies, and coordination of logistics activities.
Interventions can, therefore, affect the surgery schedule of the Lap Sponge product, and its forward-planning approach minimizes interruptions related to raw materials provided by its competitor, BKA MED. Buyers reap benefits through dependable lead time predictability. Any challenges are addressed proactively instead of reactively.
It is this kind of reliability, and not spot orders, which is the basis of long-term contracts and forms the heart of customer relations operated by BKA Med.
Partnering Beyond Transactions
BKA MED views relationships more as partnerships than as transactions. This has an impact on how the organization supports its clients prior to and following the placing of orders.
Technical documentation support, audit preparation, and assistance with product validation will also remain. These forms of technical documentation, especially for complex purchasing situations, can alleviate internal workload for buyers. A Lap Sponge is small in physical size, though with big responsibility. Likewise, BKA MED knows this well and advances support.
Let’s be clear: trust built over time is the source of long-term value in surgical supply chains. Such emphasis on transparency and responsiveness reinforces that trust.
Choosing the Right Lap Sponge Partner
While the process of finding the right sources of supply of a product such as the lap sponge is not necessarily easy, it is not simply a matter of finding a product that meets the essential criteria and passing it off as the right choice.
As this guide has demonstrated, there is much more going on behind the scenes, including considerations such as materials, processes, quality, regulatory, and supply. All of these are essential features that have a direct impact on the end-user, which is the patients receiving medical care.
That said, BKA MED prides itself on quality and supply, including end-to-end process controls, quality, compliance, and supply, especially within a surgery environment, which is what sets them apart from the competition. If you are looking to source the right quality of the product on a large scale, we at BKAMED invite you to submit a request for a quote.

