Every procurement expert has faced this difficult question before: “Can we trust this supplier in the long term?” When it comes to gauze balls, the danger usually lurks beneath a promising price and a professional certification. On the outset, many manufacturers seem pretty much the same. In reality, though, various factors in raw material and quality control could create significant after-effects.
This article has been written specifically to help procurement teams make educated choices and moves from guesses. We’ll give a detailed investigation of the top 10 Chinese manufacturers of gauze balls, describe the actual process of making gauze balls, and define the quality standards which matter in mass procurement and the verification of quality control and compliance with procedures.
The last part introduces why expert global buyers choose BKA MED as a supplier in mass markets with regulatory requirements or restrictions.
Key Takeaways
- The top 10 gauze ball manufacturers in the China are BKA MED (ranking 1st for its vertically integrated production chain, ISO 13485, CE, FDA compliance, customization for NGOs, and partnership approach to risk management), followed by Winner Medical Co., Ltd., Zhende Medical Co., Ltd., Jiangsu Jianerkang Medical Dressing Co., Ltd., and others
- Gauze balls are produced through processes of purification, carding, and alignment of raw cotton fibers, weaving into uniform gauze material, cutting and rolling into balls of specific sizes, possible sterilization, and finally inspection for absorbency and traceability.
- There are currently three ways of sterilizing gauze balls: EO (Ethylene Oxide), which is good for penetration of porous materials but needs aeration to reduce residues), Gamma Radiation (residue free but can weaken cotton), and non-sterile cleanroom controls for cleanliness in non-invasive procedures.
- The quality of cotton used in gauze balls is based on long-fiber staples for increased absorbing capabilities and reduced lint, hydrogen peroxide or oxygen bleach processes for reduced residue and use of cotton-polymers if required for added tensile strength in non-sterile products.
Top 10 Gauze Ball Manufacturers in China

Below are the leading players in the industry, chosen based on their export quantity, certification requirements, and manufacturing capacity. The companies mentioned below form the backbone of the worldwide supply chain for quality Gauze Balls and are what procurement managers need to stabilize contracts with suppliers.
1. BKA MED (Hubei Baikang Medical Instrument Co., Ltd.)
Hubei province is commonly known to be the core for China’s medical gauze, while BKA MED (Hubei Baikang Medical Instrument Co., Ltd) is among the major players in this area. This company operates in a vertically integrated setup where the whole production process is closely managed from the very initial stages of weaving to the inspection stage. This is rather beneficial for the distributors as they want all the Gauze Balls issued to them to have the same standards.
● Production Capacity and Customization
BKA MED has its own weaving, bleaching, and finishing processes in its large facilities through its company, BKA MED MANUFACTURING. Through this approach, it is able to control and manage its mass production in a way that is ideal for large contracts and with scalability in its production for a long-term contract for bulk production. In this way, it is able to eliminate any quality variations in its production, which could be experienced when it outsources its production to another company for its textiles.
The company also has a vast OEM as well as ODM to cater to institutional buyers. The flexibility thus ensures that healthcare providers can get their requirements fulfilled for specific surgeries or clinical needs. Whether such requirements are for custom sizing or custom packaging for NGO kits, the manufacturing capacity is capable of delivering such custom requirements without any delays.
● Key Certifications
Compliance is a non-negotiable for BKA MED, and its adherence to international standards is evident from its ISO 13485 certification, which is the highest standard for quality management systems for medical devices. It, therefore, ensures that each and every stage within its production cycle is documented and auditable.
Additionally, the company has CE marking status as well as FDA registration; thus, their product can be used by both the European market as well as the United States market. In the perspective of a procurement officer, such markings form the minimum point of trust that the Gauze Balls product has met all the necessary testing required by health authorities across the world.
2. Winner Medical Co., Ltd.
It has been reported that Winner Medical is among the largest and most influential players in the Chinese market in terms of manufacturing medical consumables. It is based on their comprehensive industrial chain, which begins with the selection of cotton. Their huge size enables them to provide for the top levels in the healthcare market with outstanding efficiency.
● Strengths in Bulk Supply
Being a publicly listed company, Winner Medical conducts business operations that entail a high level of transparency and stability. This is important to institutional buyers who wish to engage a supplier qualified to execute enormous contracts for supplying a bulk of their needs for several years. The production plant has massive automated production lines that ensure high production of Gauze Balls with little human error.
The internal processes in Winner Medical are set up to surpass the essential standards required by the regulatory bodies. The company spends substantial amounts on research and development, most in the non-woven, woven, and cotton technologies sector. This enables them to ensure that the absorbency features and durability of their products are the leaders in the market.
● Global Reach
The distribution network reach of Winner Medical covers over 100 countries, making it a truly global organization. With this broad reach, it is evident that this organization is well aware of all local nuances. Purchases would also benefit from a long history of supplies to major international medical establishments.
This worldwide experience means a smooth shipping and processing procedure. Then, when purchasing a considerable quantity of Gauze Balls, a company’s capacity to comply with international trade rules is as significant as a product’s quality. However, Winner Medical has demonstrated its capacity to handle this for a long time.
3. Zhende Medical Co., Ltd.
Founded in 1994, Zhende Medical has expanded to become a prominent manufacturer of healthcare consumables. The company has been focusing on the development of a comprehensive global distribution network that meets the varied demands of healthcare systems and medical distributors.
● Company Background
The years of experience that Zhende Medical possesses mark a rich knowledge base in medical textile manufacturing. The years that have gone by have allowed Zhende to streamline its operations to maintain high-speed production while incorporating sensitive requirements in wound care items. Such experience rates high on the parameter of institutional knowledge as compared to other new factories.
Even though the company involves itself in the mass production of numerous goods, its gauze-based wound care products are an essential commodity within its exports trade. The facilities are designed to maximize mass production so that institutional purchase orders for Gauze Balls are ensured to be fulfilled regardless of the market demand at that particular stage.
● Regulatory Footprint
Zhende Medical holds registrations for ISO, CE, and FDA within its respective categories for different products. This makes the import process easier for buyers in regulated environments. It is important to note that Gauze Balls are often included under textile products; however, Zhende is renowned worldwide for its expertise in gauze production.
Quality commitment by the company can be seen through its adherence to international testing standards. Each consignment undergoes extensive testing to ensure that it is free of contaminants and that it satisfies the criteria of absorbency. This attention to detail makes Zhende a preferred supplier in government tenders as well as institutional orders.
4. Jiangsu Jianerkang Medical Dressing Co., Ltd.
Originating in 1993, Jiangsu Jianerkang Medical Dressing Co., Ltd. (hereinafter referred to as Jianerkang Medical) was founded as the Jintan County Zhixi Sanitary Material Factory. It has grown to be one of the largest professional medical dressing manufacturers in China. In November 2024, the firm reached an important milestone: listing on the Shanghai Stock Exchange, which underlined its market-leading position.
● Integrated Manufacturing Excellence
Jianerkang operates on a large scale, covering areas up to 350,000 square meters and having over 1,800 employees. They have more than dozens of automated high-temperature and high-pressure bleaching lines. With such technology, each lot of Gauze Balls can be produced with the highest purity and absorbency to meet the demands of highly demanding surgical theaters.
Jianerkang’s line of gauze products has been engineered with superior structural integrity related to wound care. As an industry leader in wound care dressings, they supply a broad portfolio of dressing types to ensure that clinicians have exactly what they need during every stage of a patient’s recovery.
● Global Reputation and Research and Development
The firm has managed to pass various on-site inspections conducted by the U.S. FDA with flying colors, most of which have yielded zero defect outcomes. Their regulatory success can be attributed to their sophisticated testing center that attends to both physical and chemical testing. This ensures that the firm’s products continue to be safe for use in the toughest medical settings across the globe.
Jianerkang has an international presence that stretches across America, Europe, and Africa, where they are recognized as a company that has fast delivery and top-quality goods. They possess specifications that exceed 2,000 product specs, which is quite a unique characteristic since most companies, especially in the medical textile industry, fall short of this milestone.
5. Hubei Qianjiang Kingphar Medical Material Co.,
Kingphar is a group business that focuses on integration in weaving, bleaching, and manufacturing finished products. It is an “all in one” business primarily located in Hubei, which is a province famous for the superiority of the cotton it produces.
● Production Specifications
The company, which was established back in 1989 specializes in 100% cotton Gauze Balls and offers them in several sizes and ply options. This is important for institutions that have to stock different versions of the product for specialized tasks in surgery. Whether the requirement is for sterile or non-sterile variants, Kingphar has the capacity to produce both on a large scale.
The use of high-grade cotton yarn ensures that the resultant gauze is soft, super-absorbent, and free from loose fibers. These specifications are important to prevent complications during surgical procedures and wound cleaning, where linting can cause big problems in patient recovery
● Facility & Compliance
Kingphar companies comply with GMP (Good Manufacturing Practices), which ensures that the entire production facility is strictly controlled for quality and purity of production. The use of Clean Rooms with classification of up to Class 100,000 for final product production and packaging is their hygiene standard.
Such controlled environments play critical roles in creating sterile Gauze Balls that are recognized by international safety standards. With high-tech setups combined with ISO and CE procedures strictly followed, Kingphar Company emerges as one of the best suppliers for healthcare systems that put safety before everything.
6. Hubei Zhongjian Medical Supplies Co., Ltd.
The company, Hubei Zhongjian Medical Supplies Co., Ltd., was founded in 2005 and has rapidly become one of the main strengths within the Hubei medical textile industry. The company has a registered capital of 56 million yuan and employs 700 staff, who are known for its massive production capacity. The company now produces medical gauze with an annual output of 250 million meters, which can be deep-processed into different specialties.
● Advanced Production and Scaling
The company has two factories that employ a modern assembly line system. This combines grey cloth weaving, degreasing, bleaching, and automatic packing. It would be very useful to have such an optimized process in order to meet large-scale hospital contracts and medical stock requirements in producing Gauze Balls.
In the medical field, there commonly arise the issues of how to avoid the adherence of gauzes to the surfaces of the wounds. These issues have been addressed by the brand Zhongjian through the creation of different non-adhesive components with high absorbency in their product range. This element of considering the demands of the final application of the product makes their medical supplies ideal for trauma centers.
● Compliance and Economic Impact
Since its inception, the company has enjoyed very conducive working environments, having passed ISO 13485 and CE certifications in its first year. This transparency and emphasis on quality has made them “contract-honoring and trustworthy” in their province, Hubei. This reputation also stems from a very sound financial position, as they make substantial annual sales as well as tax payments.
Apart from the capabilities for manufacture, they have been known for non-standard and customized products for meeting specific customer needs. This aspect is important for B2B customers for meeting specific counts or weights for Gauze Balls.
7. Suzhou Sunmed Co., Ltd
Suzhou Sunmed has over 30 years of experience in the medical consumables field. They are based at the industrial center of Suzhou and have utilized their geographical advantages to create a sound manufacturing and export business. They produce a wide range of items; however, their core concentration remains on cotton medical supplies.
● Production Footprint & Markets
It has various production facilities that enable a diversified production strategy. This benefits a constant exported shipment to Europe, Africa, and Asia in over 60 countries. With various facilities, Suzhou Sunmed can easily minimize local production risks for a continuous supply of Gauze Balls to its international partners.
The factory has the ISO 13485 certification and CE mark, which satisfies the basic requirements for medical products in the European medical sector. This regionalization enables them to design products that fit the preferences of the European medical sector.
● Product & Quality Strengths
The FDA registration of product lines of the company, Suzhou Sunmed, is another attribute that makes it an appealing choice for North American buyers as well. It is famous for its consistent standards of exports, an aspect which is crucial for maintaining brand reputation in the B2B market.
It seems that the Gauze Balls produced by this company are trusted by institutions for having the right price for the quality offered. This firm has many years of experience in its industry and is able to source its raw material inputs in the most optimized manner, and still maintain its high quality since the final product is of medical quality.
8. Yangzhou Goldenwell Medical Devices Co., Ltd.
Having been in business since the late 80s, Yangzhou Goldenwell is among the relatively established brands in the Chinese healthcare device market. It has taken the company decades to establish a balanced product line that encompasses various consumables for wound care, as well as various medical instruments.
● Company Longevity & Range
The longevity of Yangzhou Goldenwell is a demonstration of the company’s adaptability to a dynamic market and environment. For a procurement manager, it is a positive signal that expresses both reliability and continuity of the kind that is a priority for high-stakes supply chains. This continuity encompasses different sizes of Gauze Balls.
They have a reputation for being a “one-stop shopping” supplier for many distributors. Since manufacturers provide everything from bandages to syringes, this allows customers to consolidate their delivery and purchasing needs with them. It could provide them with a huge benefit regarding logistics and quality checks.
● Export Experience and Certifications
With ISO, CE, and FDA certification, Yangzhou Goldenwell is prepared for international trade. They also have flexible customization capabilities, especially helpful for bulk buyers with certain label or packaging needs for the respective markets that they are catering to.
The experience of the company in processing large exports ensures that all documents accompanying the exports are accurate. This helps avoid possible clearance delays when exporting the Gauze Balls to health facilities where they are most required.
9. Nantong Jianan Medical Products Co., Ltd
Nantong Jianan Medical Products Co., Ltd. is a private enterprise that has been engaged in the medical supplies business since 1999. Based in the Nantong Economic Development Zone, they have been perfecting an in-house manufacturing process that spans the stages of spinning and weaving to bleaching for the past couple of decades to meet the demand for Gauze Balls for their various international clientele.
● Factory Capabilities and Market Reach
The company has an equipped clean workshop and a workforce of more than 200 skilled personnel. This enables the company to meet the high volume requirements while still being precise with the technical skills needed for the production of the surgical materials. In the recent past, they have been able to reach markets in over 50 countries; they are a household name within the European and Southeast Asian markets.
They have a broad product line, but they are most renowned for their continuity in weaving textile products. A procurement manager would find it important to understand what is a gauze pad and what is a folded ball, and indeed, Jianan is capable of providing both products in multiple ply and sizes, which would ensure that a hospital does not look for multiple sources for a variety of absorbents.
● Quality Standards and Compliance
Jianan Medical prioritizes quality over profit, a code that has allowed them to grow steadily over the years. The company is ISO 13485, CE certified, and they also maintain an active FDA registration. Such certifications ensure that their Gauze Balls comply with the strict standards set by worldwide medical organizations regarding safety.
By locally producing products near the Shanghai Port, they also provide substantial logistics advantages, especially in terms of bulk exportation. In this way, they are also able to limit transport time as well as export costs in accommodating high-volume buyers.
10. Zibo Eastmed Healthcare Products Co., Ltd.
Zibo Eastmed is a specialized manufacturer and has created a prominent niche for itself in the medical textile industry. It is mainly catering to the overseas markets in Europe, Africa, and the Middle East, which have different requirements for their respective markets.
● Product Range & Export Reach
The company offers an extensive range of gauze-based medical textiles, including Gauze Balls that are an integral part of their export offerings. Their clients from all over the world benefit from their flexibility to customize products according to institutional requirements. They know that a European hospital may have different packaging demands compared to a Middle Eastern clinic.
This adaptability is coupled with an efficient logistics system. Zibo Eastmed has developed a shipping network that guarantees their products reach different destinations of the world quickly. For a B2B perspective, this is an important aspect that reduces the time taken for delivery and ensures there is adequate stock.
● Quality Certifications
The ISO and CE certification is a crucial part of their business models. The certification enables them to access tenders from other countries as well as to serve healthcare providers who demand quality control evidence.
The production process in Zibo Eastmed involves a number of control points, which are aimed at ensuring that the Gauze Balls achieve the required level of absorbency and purity. This is important for helping to win the confidence of institutional buyers, who are unable to afford to take any chances regarding the equipment they use in their work.
What Are Gauze Balls in Clinical Procurement Terms
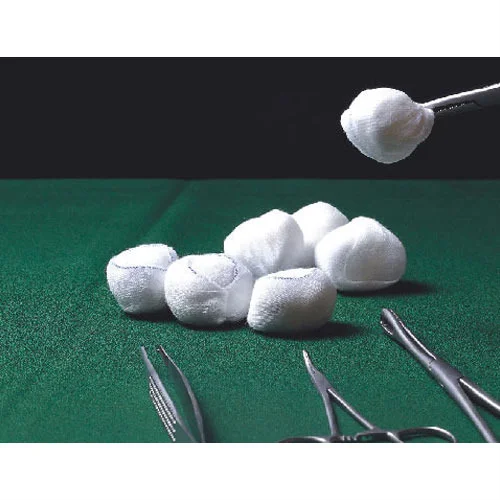
In institutional procurement, Gauze Balls are defined as standardized, disposable, absorbable, & multiple healthcare-related usage–based medical products. Gauze Balls are not regarded as secondary, additional products; rather, these products are ranked as essential items within wound care and surgical supply categories. The rationale for buying these items in bulk is linked to the foreseen demand for these products.
In hospital setups, Gauze Balls have been included in purchase catalogues and procurement software under various categories like wound care and minor surgery items. This is to enable standardization of dimensions, number of layers, sterility status, and packaging types. In NGO procurement tools, they have also been included in emergency sets, mobile clinic setups, and humanitarian response boxes.
Classification-wise, Gauze Balls occur at the intersection of wound care and surgery preparation supplies. Gauze Balls are used both in conjunction with wounds and in conjunction with preparation and cleaning processes. This accounts for their presence in the budgets of multiple departments instead of being under the control of just one.
There are some differences in the acquisition that can affect the evaluation of Gauze Balls:
- Sterile vs Non-Sterile:Sterile variants have higher priority for operations and invasive procedures, while non-sterile variants can be used for cleaning and external wound treatment.
- Absorbency vs Unit Cost: Absorbers with high levels of absorbency may be less material-intensive per procedure; nonetheless, high volumes may outweigh these savings with unit pricing.
- Volume Demand Rates: The rates of volume consumption may tend to be considerably high at the emergency and surgery departments.
Difference Between Gauze Balls, Gauze Swabs, and Gauze Pads

Though these items are often packaged together, Gauze Balls, swabs, and pads all have different purposes. From a physical perspective, Gauze Balls take on a rounded form that will enable appropriate absorption. Swabs typically take on a folded square design, while pads take on a flat and bigger design.
The efficiency of packaging and dispensing is another crucial area that makes institutions distinguish these products from others. Gauze Balls are easy to hold by forceps. The Gauze Balls can be dispensed one by one from a pack, which reduces wastage in fast-track settings. However, Swabs and Pads are usually dispensed in a definite number, which may not necessarily correspond to the required number for a certain procedure.
Substitution from a chain perspective may not be that easy. Though technically Gauze Balls can substitute Balls in certain applications, substitution may influence work-flow efficiency and usage patterns alike. Hospitals trying to achieve SKU simplification may analyze applications where Gauze Balls offer certain advantages in functionality to be retained as distinct SKUs rather than including all products under one category.
This difference also assists in explaining what a gauze pad is as opposed to a ball. The gauze pad is normally flat and stratified, designed for wound coverage rather than cleaning, dabbing, or fluid absorption.
Core Institutional Applications
● Surgical & Operating Room Use
In operating room situations, Gauze Balls work best for absorbing blood and maintaining good visibility. Gauze Balls also work well in handling tissues. An advantage of these Gauze Balls is that they provide good control due to their shape.
● Wound Care & Dressing Preparation
In the outside environment of the operating room, Gauze Balls has subsidiary involvement related to wound care processes. These materials are often employed for cleansing wounds, disinfection by antiseptic treatment, as well as preparing the area for dressing application. It is here that general wound dressing types are introduced, where gauze products serve as the principal base for various treatment regimens.
● Emergency & Trauma Response Kits
Emergency centers and trauma centers have been relying on Gauze Balls. These Gauze Balls can be easily set up and do not need training. In a crisis or emergency situation, products that do not need many preparations and training can be considered ideal. These items can also be placed in crash carts.
● First Aid Kits for NGOs & Field Hospitals
NGOs and field hospitals appreciate Gauze Balls for their versatility. Gauze Balls can be utilized for both first aid and basic procedures, as well as wound cleansing. The product being included in standardized kits makes training and handling easy, particularly in resource-poor settings.
Why Gauze Balls Remain a High-Volume Consumable
Despite technology improvements for wound treatment, Gauze Balls top hospital purchase lists. This is due to a couple of factors.
- They are economical. They are affordable in multiple quantities from trusted manufacturers per use and still fulfill medical needs.
- Multi-departmental usage. Not many medical disposables can be utilized as widely across surgery, emergency, and outdoor departments. The universality of their usage leads to very high usage numbers, making the Gauze Balls a very stable purchase.
- Storage and transport efficiency. They are lightweight, compactable, and easily palletized, making storage and transportation from aid agencies and hospitals cheaper. Their long shelf life helps in bulk purchases, allowing stockpiling in hospitals and aid agencies with little need for frequent replenishment.
- Low training requirements. Gauze Balls are easy to use, which means that no specialized training is required for operating them. In terms of contracting, this reduces the indirect cost of such products to an organization. Currently, due to the challenges being faced by value chains at an increased level, products that are easy to use, scalable, as well as dependable will surely be indispensable products within those chains.
Materials Used in Gauze Ball Manufacturing
Here are the materials used in gauze ball manufacturing:
Medical-Grade Cotton as the Primary Raw Material

The key material for Gauze Balls production is medical-grade cotton. In institutional purchases, cotton is chosen not merely for its traditional usage. Rather, it is chosen for its ability to ensure a perfect amalgamation of absorbency, softness, biocompatibility, and market acceptance. In procurement terms, the superiority of cotton becomes decisive for the ability of Gauze Balls to function equally well even in millions of units.
● Cotton Fiber Length & Absorbency Metrics
The length of fibers is very essential for the fluid absorption properties in Gauze Balls. When the fibers are longer and are made of cotton, they are able to interlock properly during their processing in order to form a solid material that can hold fluid without disintegrating. Short fibers might look good at first sight, but they are likely to come apart when in contact with fluid and produce lots of lints.
The metrics of absorbency generally fall under fluid uptake per gram of cotton. For the practical application, greater absorbability means fewer Gauze Balls are required per procedure, which further dictates consumption rates at the department level.
Makers serving institutional markets typically test absorbency at various points in the manufacturing process, not just at the finished product level. A raw cotton is always prescreened before processing to minimize variability downstream. One reason established suppliers keep output stable from lot to lot, regardless of swings in the price of cotton, is this upstream control.
● Bleaching Methods and Residue Control
Bleaching is a necessary treatment to prepare cotton for use in medical applications, but the method counts. Most medical-grade Gauze Balls are compliant through reliance on an oxygen-based or hydrogen peroxide bleaching process rather than one that relies on chlorine-based materials. These methods can provide the necessary white color while keeping the amount of chemical residue left behind minimal.
Residue control is of even greater concern to buyers serving the surgical and wound care market. The presence of even trace residues of bleaching agents can lead to irritation or compromise healing. From the point of view of compliance documentation, residue analysis is often part of the quality dossier submitted to the hospital buyers.
However, the issue that sourcing discipline emerges to address is the amount of residue that remains after the bleaching process. The companies that are trying to optimize the process by getting the cycles out the door quicker are the ones that end up having difficulty controlling the amount of residue that is left behind. This, in the end, affects the products. Those that are bulk purchasing the products are not necessarily concerned with the chemical dynamics behind the residue, but rather the consistency of the suppliers.
● Whiteness Index & USP Standards\
The whiteness of Gauze Balls also serves other purposes. The whiteness of Gauze Balls is measured using indexes. The indexes aid in creating product consistency. The whiteness index facilitates medical professionals to detect blood, exudates, or contaminants.
In lots of export countries, especially North America, USP specifications must be met. USP provides acceptable levels for purity, whiteness, and performance levels and factors. Suppliers making Gauze Balls intended for hospitals in other countries usually normalize their manufacturing specifications around USP levels or around pharmacopeial levels even if not mandated.
It is a proxy indicator for procurement officers, while a lack of consistency between shipments could indicate problems with control or processing of the materials if it fluctuates widely between shipments.
Alternative or Blended Materials (When Applicable)
While medical cotton is still the preferred material, there are Gauze Balls that use other types of fibers. These types of fibers are normally incorporated in response to functionality or availability issues, rather than advancements in medical capabilities.
Cotton-polymer blends: There is a rare occurrence of Cotton-polymer blends being used in Non-Sterile Gauze Balls that are meant for purposes of cleansing and preparation. This is due to the added advantage that the synthetic material offers in increasing tensile strength while minimizing linting. However, the absorbency is lowered.
Some cheaper variants include rayon blends. This is because rayon is capable of imitating the feel of cotton, but such potential comes with risks associated with its use. This material has been shown to be inconsistent in moisture and susceptible to degradation during certain sterilization processes. Institutions are less concerned with savings on such potential risks.
The catch is that alternative materials aren’t necessarily unfit for use, although they require stricter specifications. Organizations like hospitals and NGOs that allow blended Gauze Balls would have some very limited applications and would depend on documents provided by suppliers for Gauze Balls performance.
Why Material Quality Shapes Long-Term Supply Performance
The material choices that were made at the point of cotton selection have an undeniably downstream effect that spreads across the entire SLS cycle for Gauze Balls. They affect absorbency rates, the ability to be sterilized, shelf-life compatibility, and even packaging efficiency. In this case, for the bigger consumer, they get fewer surprises.
In mass purchasing, consistency is more highly prized than incremental gains in cost savings. Gauze Balls with consistent performance levels, regardless of the department, shipment, or year, simplify administrative and clinical processes for the institution. It is for this reason that more experienced purchasing personnel consider material strategy, in addition to certifications and price, today.
As the world’s healthcare systems continue to stress on reliability and compliance, the need for material transparency is expected to continue being a defining characteristic. In this respect, cotton is more than just a raw material. It is the cornerstone upon which the trust for the Gauze Balls production process is based.
Gauze Ball Manufacturing Process (From Fiber to Finished Product)
Knowing how Gauze Balls are made is important for institutional purchasers who want to evaluate the factors involved beyond the actual price of the product. This is because every stage of product development is a potential source of control or risk, depending on how it is managed for the procuring entity.
This section describes in detail the entire process from raw cotton handling to final delivery of the finished product that is ready to be shipped to the hospitals and NGOs in a simplified manner.
Raw Cotton Processing
The process of the production of Gauze Balls and how they come into being begins long before the actual process of weaving and rolling. The initial process is preparing the raw cotton for medical purposes.
● Cleaning, Carding, and Alignment
The raw cotton will come with inherent contaminants like seeds, vegetative particles, and oils. The beginning process will involve cleaning, either mechanical or chemical, where loose particles are removed, and the fibers are treated to soften them. However, the cotton will need to go through the carding process, which will align the fibers in a uniform web.
Aligned fibers are paramount. This presents a better gauze when the fibers are aligned in one particular direction. This impacts the Gauze Balls in that it relates to how the gauze holds up in a wet situation as a ball.
In this stage, the quality control process begins for the manufacturing process itself. The density and orientation of the fibers are inspected for predictable performance in later processing operations, an unstable process causing difficulty or defects later on being difficult to reverse.
● Contaminant Removal Controls
Medical cotton requires high purity. Besides the physical debris that can be viewed, other possible debris could be oils, dust, and other fibers during the harvesting and packaging process. High-end companies produce medical cotton using filtration systems and wash processes.
For institutional buyers, contaminant control is not a quality concern but a reality. For instance, pure cotton prevents Gauze Balls from being used in an operating room or wound care center where it causes irritation or infections.
This is where an important difference becomes apparent too. It is not uncommon for procurement teams to ask if one can use cotton balls instead of gauze under certain circumstances. In terms of manufacturing, the answer has to do with processing strength. Cosmetic cotton balls may not have the same level of contaminant safety or preparation for an actual procedure, which is why Gauze Balls are the choice of institutions.
Weaving & Gauze Formation
When the cotton fibers are cleaned and aligned together, they turn into a gauze fabric. This is where the overall physical characteristics of the Gauze Balls are determined.
The thread count and mesh control are important parameters. The thread count is the number of threads per unit area, whereas the mesh values the openness of the fabric. The balanced mesh enables fluid to pass through with stability.
If it is too loose, it can easily rip or fray apart. If it is too dense, it will become non-absorbent. The count range of a Gauze Ball made by a manufacturer aiming toward worldwide sales is standardized according to their internal requirements, meeting the requirements for pharmacopeias.
Weaving is followed by uniformity testing, which is done to confirm consistent mesh, weight, and strength through samples taken from different sections of the fabric roll. This is because Gauze Balls are cut and rolled from large sheets of fabric. Inconsistency here gets multiplied into thousands of units.
For procurement teams, weaving quality explains why two visually similar Gauze Balls can perform very differently in practice. Uniform fabric ensures predictable absorption and handling across departments and shipments.
Cutting, Rolling, and Ball Formation
The conversion comes right after the production of gauze fabric into finished Gauze Balls. This stage brings about precision combined with efficiency, especially during high-volume production.
Automation levels will vary from one manufacturer to another. Fully automated lines cut the gauze into very specific dimensions, roll it into consistently sized balls, and then compress them so they do not lose their shape. Semi-automated or manual processes rely more on operator skill, hence variability.
Here, size tolerance standards become critical. Normally, Gauze Balls are specified by diameter and weight. Small deviations in either respect can affect absorbency and packing counts. For institutional buyers, size consistency provides predictable consumption rates and simplifies inventory planning accordingly.
The vendors supplying hospitals and NGOs set a certain tolerance limit, and they strictly monitor it. The balls that do not come under the set limit are sorted out before packing. This helps avoid any complaint and returns purchases in case of bulk supplies.
This stage also impacts the usability aspect. Properly formed Gauze Balls are very easy to handle using forceps and retain shape during handling and application. Improper methods of rolling cause them to distort or unravel, thus hindering the healthcare process.
Sterilization (If Applicable)
It is not a requirement for Gauze Balls to be sterilized, but if this is done, then sterilization becomes one of the most precisely controlled steps in this process. It is important to consider a method compatible with cotton.
● EO Sterilization
Ethylene oxide (EO) sterilization is commonly used for Gauze Balls because it is able to easily penetrate porous surfaces. It is most appropriate for products that have complex packaging designs.
However, EO requires effective management of the residues. After the sterilization process, there is a need for aeration to eliminate any remaining gas. The industry has documented various aspects of the process, considering that the residues are strictly controlled by various countries.
For the procurement side, the flexibility of EO sterilization requires good process control. Suppliers can offer validation reports and batch records to prove their validation.
● Gamma Sterilization
Gamma radiation has high energy that kills microorganisms. The method is effective with no chemical residue left, which attracts quite a number of customers. But sometimes, the radiation can affect the strength of cotton.
It is essential for the gamma producers to validate the dose levels in Gause Balls, which will help in retaining the integrity level. It is usually preferred for pre-pack units instead of bulk units.
● Non-Sterile Production Controls
Even for Non-Sterile Gauze Balls, strict cleanliness practices must be implemented. Cleanroom technology and air handling, combined with cleanliness practices for personnel, are used to keep bioburden down.
These are relevant because non-sterile products have many applications in wound cleansing. These can easily get contaminated due to poor hygiene practices in production before they are even sterilized.
Comprehending these variations enables the consumer to match product requirements with actual application requirements, such as how not to have gauze adhere to the wound, which is contingent on downstream applications of dressing rather than on the process of sterilization.
Final Inspection & Batch Release
Prior to Gauze Balls being cleared for dispatch, final inspections are done, and batches are released. This is the final safety check to ensure that defective Gauze Balls are not delivered to institutional buyers.
Optical inspection involves looking for any signs of discoloration, distortion, and presence of foreign bodies. Although this is a very simple process, it still identifies defects which might not have been picked up by automated equipment. Inspectors assess samples for each batch of merchandise, depending on predetermined criteria for acceptability.
The absorbency tests are also conducted for the completed Gauze Balls. They are weighed, immersed, and then weighed again to ensure that they can meet the internal standards. The reliability of absorbency enables consistent performance when used for various medical purposes.
Traceability documentation is the final process in which a unique identification number is given for every batch, tracing it to raw cotton, processing conditions, and test reports. This process is extremely useful in hospitals and NGOs that import in large volumes.
Quality Control Standards for Gauze Balls in Bulk Procurement
In institutional procurement, quality control is where product claims are either validated or invalidated. For Gauze Balls, quality does not reside in its appearance. It is assessed for its repeatability, demonstrated by systematic testing and controlled release, where each item shipped performs exactly like its predecessor.
These controls help hospitals, NGOs, as well as government purchasing bodies, minimize clinical and administrative risks. When Gauze Balls are procured in bulk quantities, even a slight discrepancy in quality can become a significant problem. This is why the criteria for quality controls are as important as any certification or price.
In-Process Quality Checks
Quality checks are done while Gauze Balls are being made and not after they are made. Quality checks aim to eliminate flaws before they occur rather than after they are completed. Viewing this process from a procurement stand, this is a sign of an effective quality system.
While raw cotton is being processed, manufacturers check the purity, moisture content, and orientation of the fibers. If anything goes out of hand here, problems may be experienced with absorbency as well as strength properties down the line.
When gauze fabrics are woven, thread count and mesh uniformity are monitored on a constant basis. Robotic sensors or predetermined periodic checks are used to ensure that fabric density measures within predetermined parameters. In the case of Gauze Balls, this helps ensure consistent absorption and durability as the fabrics are processed into rolls.
The conversion stages, whether it is cutting and rolling, for example, are also closely watched for any deviations in size, weight, and shape. It is very relevant to note that Gauze Balls with varying sizes could pose problems regarding packaging and consumption in institutional settings.
In-process checks decrease the need for end-stage rejection. They offer the data that the quality teams can utilize to uncover trends before they turn to problems. For bulk buyers, it allows for fewer surprises and more regular deliveries for long-term contracts.
Finished Product Testing
Final product tests represent the final approval that Gauze Balls comply with specified requirements. Final product tests differ from in-process checks in the matter that they confirm requirements for a product as it is finally delivered and utilized.
● Absorbency Rate Testing
Absorbency is among the most important evaluation factors for Gauze Balls. This is usually done by determining the amount of liquid that can be absorbed by the sample compared to its weight. This information is useful in ascertaining that the item can work well under surgical, emergency, or wound treatment conditions.
It makes the product less likely to be overused during procedures. As for the purchase perspective, the benefits include the ability to estimate consumption correctly.
● Lint & Particle Shedding Tests
Linting is a known issue associated with gauze-based medical supplies. Excessive fiber sloughing may be a hindrance to processes and may make caring for the wound more difficult. Testing of Gauze Balls includes agitating a sample and assessing particulates.
Low lint means the fibers are good and processed well. This will help hospitals and NGOs to minimize complaints regarding the use. It will enhance the confidence level regarding the use of fibers on a large scale.
● Microbial Load Testing
The Gauze Balls, even if non-sterile, must comply with certain microbial limits. The microbial load test is done to determine the viable microorganism level on a finished product before it undergoes sterilization or its use.
This is because the test procedure is able to establish the effectiveness of the hygiene control in the manufacturing process by positively identifying the presence or absence of these standards. In institutional purchasing, this is assurance that products reaching healthcare settings meet minimum standards on safety.
The testing of the end product is normally conducted on a statistically significant sample drawn from among every batch. The information obtained is also documented and retained within the batch record.
Documentation Buyers Should Request
Quality testing is only beneficial as a service if it is supplemented by credible documentation. In bulk purchasing, documentation can be as valuable as the items themselves. Quality documentation is important for confronting challenges of compliance and audits, which can be posed by the supply of Gauze Balls.
- Certificates of Analysis (COA) provide a summary of the major test results. These usually consist of absorbency data and microbial limits. A valid COA establishes performance features and test results rather than specifications.
- Lot traceability documentation enables the tracing of each consignment of Gauze Balls from the raw materials to the dates of production and testing documents. Lot traceability is crucial in cases of recalls, investigations, or regulatory examinations. It is also used during quality audits within a hospital set-up.
- Sterilization validation documents will be required for sterile Gauze Balls. These documents will show that the sterilization process is validated, checked, & consistently performed. Cycle parameters, biological results, & residue data will be provided in these documents.
- Change control notices receive little attention but are very critical. They capture all changes related to materials, processes, and equipment that might have an impact on product function and performance. Regarding long-term contracts, change control transparency enables purchasing personnel to determine whether modifications pose new risks to an enterprise.
Let’s be clear: documentation is not a matter of administration. It’s a tool for managing risks. Contract manufacturers who are proactive about supplying well-organized documentation generally adapt more easily to the institutional system for providing supplies.
Why Quality Control Determines Long-Term Supplier Value
Quality control requirements influence the performance of Gauze Balls not only once but on a constant basis over a period of time. For a bulk buyer, durability eliminates the cost of complaints, retraining of workers, and emergency measures required from resources.
Effective checks in the process prevent variation before the final products are produced. Effective testing ensures that products are as expected when they are delivered. Documentation helps in ensuring compliance with regions or regulation systems.
In large-volume purchasing, it is predictable because of all those factors mentioned above. Gauze Balls supplied by manufacturers with well-controlled processes for quality perform in predictable ways regardless of purchasers, business units, and applications.
With the rise of scrutiny of healthcare systems and NGOs, the need for quality control has ceased to be a point of differentiation. It has become the minimum need. Entities that understand that it is the minimum need are better equipped to assist institutional buyers.
In this regard, assessing the criteria for quality control is not a process of searching for perfection. Rather, it is a process of finding partners who can supply reliable Gauze Balls in volume.
Global Compliance, Certifications, and Regulatory Pathways
In the case that Gauze Balls are acquired for institutionalized usage, non-compliance is not merely a requirement to go through, as this is basically the system that enables products that are produced in another country to be used without posing any possible health risks or legal problems along the way. In institutional facilities such as hospitals or NGOs or centralized procurement offices that import in bulk.
This section outlines the principal worldwide compliance considerations relevant to Gauze Balls and primarily addresses those matters of which purchasing organizations must be aware when considering manufacturers for Europe, North America, and nongovernmental organization-driven markets.
ISO 13485 and Manufacturer Selection
ISO 13485 is generally considered a baseline quality management system for manufacturers of medical devices. In terms of Gauze Balls, a product that generally qualifies as a low-risk medical disposable, ISO 13485 does not in any way alter the usage process of the product.ISO 13485 improves how the product is manufactured, documented, and maintained.
For a buyer, the existence of ISO 13485 shows that the manufacturer has a quality system in place. A quality system consists of procedures, risk, and corrective measures, and it is essential, especially when High Volumes of Gauze Balls are delivered for a longer period of time in a contract.
ISO 13485-certified manufacturers must also be able to systematically control changes. Changeover in any type of raw material, equipment, or processes must be reviewed for its effects and documented. For hospitals and NGOs, it ensures that unexpected variations in performances after deliveries do not happen.
It is also important to recognize that the ISO 13485 certification pertains to the manufacturing site level, not the product level. Procurement teams with requirements for Gauze Balls should ensure that the certification includes coverage for the specific factory producing the goods and not depend on corporate-level claims of certification.
In practice, ISO 13485 eases downstream compliance. It aids in CE marking, FDA registration, and tender eligibility through a recognized quality bedrock. For purchasing institutions buying Gauze Balls internationally, it acts as an initial filter for supplier credibility.
CE Marking for European Imports
CE marking is a legal requirement to enter the European market for Gauze Balls. It confirms that the product meets applicable EU safety and performance requirements and can be freely marketed within the European Economic Area.
● MDR Classification
Under the Medical Device Regulations (MDR), Gauze Balls can generally be considered as a Class I device where they are non-invasive and non-active. This is because they do not come into contact with the body and do not involve any active functions.
Even though there aren’t many restrictions on Class I medical products, there are still many tight standards to be met. The manufacturer must set proper regulations for using Gauze Balls in order to guarantee that they function properly within those limits. There might be inconsistencies, such as misclassified products or ambiguous statements of intent.
In regard to acquiring teams, knowledge of the MDR classes can benefit them by setting proper expectations. Being a Class I device makes market entry easy; however, documentation along with post-market surveillance is still required.
● Technical File Requirements
CE marking is supported by a technical file that documents how Gauze Balls are designed, manufactured, tested, and controlled. This generally includes product specification, risk analysis, considerations on biocompatibility, and quality control data.
Although the buyers themselves do not usually get the complete technical file, very often a summary or declaration is requested, confirming that it is complete. For Gauze Balls being supplied to European hospitals, this is a file the manufacturer should maintain and update when changes take place.
Here’s the thing: technical documentation is never truly static. Under MDR, manufacturers are supposed to monitor the performance of their products and take field feedback into account. For institutional buyers, this ongoing obligation means less long-term compliance risk and continued product availability.
FDA Registration for U.S. Market
In the United States, Gauze Balls are regulated by the Food and Drug Administration (FDA). They are considered to be Class I medical products, and hence they do not require any premarket approval. They come under general controls.
There is a need for manufacturers who wish to export Gauze Balls to the U.S. to register their facilities with the FDA establishment registration and list devices. This assists in identifying the manufacturers of the product as well as how the product is classified among the FDA groupings. It is a means of accountability.
FDA requirements include following Quality System Regulation principles. This is closely related to ISO 13485. In this case, buyers will benefit through alignment when considering suppliers in different regions.
FDA Registration is more significant in hospitals and distributors that are multinational. FDA Registration for Gauze Balls enables these products to be more easily incorporated into US-based chains and are less likely to raise entry hurdles at import points in the US borders.
From a risk management perspective, FDA-approved manufacturers must comply with government inspections. This adds an element of assurance to those procurement teams at the institutional level that order large quantities of Gause Balls.
NGO & Tender-Specific Compliance
Apart from the regional regulations, Gauze Balls that are channeled to NGOs or supplied on a large scale may be required to meet other regulations and or standards for compliance. Such regulations and or standards might be set by organizations such as the UN or the government, among others.
Typical requirements include compliance with WHO standards, statements about ethical sourcing, and quality documentation. In the humanitarian sector, for example, the requirements may include suppliers being able to deliver on a large volume and having fast deployment capacity.
“Tender-driven procurement further accentuates the issue of documentation completeness.” Incomplete documentation concerning certain certifications might impair qualified Gauze Balls from being considered for procurement. If an NGO has projects in different regions, standard compliance makes all the difference to them.
There is no doubt that tender compliance is not simply a function of meeting the minimum criteria. It is a reflection of a willingness to work in a structured and accountable model of supply. This is something that experienced manufacturers working in NGO supply chains understand well.
Packaging, Labeling, and Logistics for International Shipments
Concerning the packing and labeling of Gauze Balls when transported across the globe in bulk, the operational aspect of packaging and labeling becomes a serious consideration. For institutions such as hospitals and government and/or non-profit organizations, the aforementioned factors affect the cost of the final product. The following section explores the packaging of Gauze Balls for shipment and institutional storage.
Bulk Packaging Formats
The packing process for Gauze Balls concentrate packs is based upon maximizing convenience, security, and packing capacity. Not like consumer packing requirements, institutional packing focuses on maximizing units per box without compromising the cleanliness and integrity of the products.
The common package types involve inner poly-bags that are packed in corrugated boxes. The inner bags may be pre-packed in predetermined numbers for simplified counting at receiving points. In the case of sterilized Gauze Balls, validated packaging processes are used for maintaining sterility from the time of packaging until storage. Medical-grade pouches are used in packaging in these processes.
Carton strength is more important than it seems. Gauze Balls face international shipping conditions that test them through stacking pressure, vibration motion, humidity variations, and prolonged shipping duration. Sturdy carton packaging with water-proof liners protects against compression and deformation, especially for ocean shipments.
Palletization is another important factor. Cartons are normally stacked in a way that maximizes container space within defined weight constraints. This maximizes the safety of the goods from damage as well as streamlines discharging goods into receiving warehouses for NGOs operating in distant areas.
Nowadays, packaging decisions are no longer limited to logistics. They affect labor efficiency for hospitals and field facilities too. Bulk-packed Gauze Balls with ease of opening, counting, and rescaling are very helpful in reducing handling and waste in high-use settings.
Labeling Requirements by Region
Labeling is a point where manufacturing, compliance, and logistics meet. For Gauze Balls, the labels should convey essential information while still remaining compliant with the various regulations of a particular region. Mistakes at this level could lead to delays in customs clearance or rejection by procurement teams.
● Language
Language is usually dependent on the destination. Most of the European markets may require labeling in their language and even sometimes in multiple languages within Europe. Whereas, some regions permit only English labeling, others do not.
It helps institutional buyers reduce internal translation needs and enables traceability along with multinational supply chains. Clear, legible text is particularly important when Gauze Balls are distributed under one contract in several facilities or countries.
● Sterility
The sterility status shall be clearly stated on all appropriate levels of packaging. There shall be clear marking of the Sterile Gauze Balls, and sterilization method references shall be included where applicable. On the other hand, the non-sterile should also be clearly identified to prevent any misuse in sensitive environments.
This is important in a hospital setup where supplies that are sourced from different places are stored in one area. Labels of sterility clearly reduce the chances of mistakes in distribution and utilization, specifically in high-pressure departments.
● Traceability
Traceability information links each shipment of Gauze Balls to its manufacturing history. This, of course, includes lot numbers, dates manufactured, expiration dates. In regulated markets, traceability supports recalls, audits, and post-market surveillance.
For NGOs and large healthcare systems, traceability also supports accountability. In situations where products have been deployed across multiple sites, the ability to trace Gauze Balls back to a specific batch simplifies quality investigations and reporting.
Let’s be clear: labeling is not just about compliance; it is a communication tool that supports safe and efficient use across diverse healthcare environments.
Shelf Life & Storage Considerations
Shelf life becomes one of the most important considerations in large purchases of Gauze Balls, especially where entities hold buffers or emergency stock. Since most Gauze Balls used in medical purposes have a very long shelf life, it becomes feasible to purchase large quantities.
Storage conditions usually include environments that are dry, with strict temperatures and avoidance of direct sunlight exposure. Gauze Balls do not react too much to environmental factors, with the exception of exposure to high temperatures or moisture.
Packaging performance is also a factor in the shelf life of sterile products. The packaging of sterile barriers needs to maintain integrity to ensure that it remains sterilized until the time of use. This is why supplier documentation can involve packaging validation, storage, among others.
Institutional purchasers also look at storage space efficiency. Smaller cartons and a standardized size are preferable for storage. In NGOs where Gauze Balls are to be stored in the field, rugged packaging ensures the balls remain safe and sound in transit.
Here’s the thing: shelf life is more than a number on a package. It is the result of a combination of materials, package engineering, and logistics excellence.
Common Procurement Risks and How Institutions Mitigate Them
Despite having suppliers and product specifications in place, there are always risks in procuring Gauze Balls in large numbers. These risks rarely occur during the quotation process. They might occur during delivery, inspection, or when the equipment is used for a longer period in various hospital sections. It is important for hospitals and acquisition organizations to be aware of these risks in maintaining continuity in medical services.
In this section, we describe common procurement risks that Gauze Balls pose and how experienced institutions counter such risks.
Quality Drift
“Quality drift” refers to the change in the performance quality of the product over time while the specifications written down for the product do not change. In the Gauze Balls procurement process, this might appear in the form of decreased absorbency, the tendency to produce more lint, or an inconsistency in the size of the final product.
The cause can reside “upstream.” Raw material sourcing, speed of manufacture, or staff level may offer subtle variables that affect output. Manufacturers without vigorous process controls are not aware of the variables until the product enters the clinical setting.
Quality drift is addressed by institutions by requiring data on the Gauze Balls tested at the batch level as well as by retaining samples from previous shipments that are taken from the earlier Gauze Balls approved for comparison to later Gauze Balls to check if they are within the approved qualities upon re-validation of their performance. Long-term contracts are usually provided to vendors that contain quality terms for re-examination if a quality drift has occurred.
Certification Misrepresentation
A problem that is often underestimated is that of certificate fraud. Some suppliers offer expired certificates, certificates of trading entities instead of factories, and/or certificates that do not match the supplied products.
Concerning Gauze Balls, it is an especially important consideration because it is a low-risk device. The ease of regulatory compliance might lead to some manufacturers finding ways to cut corners, particularly in competitive markets.
These risks can be managed through certification verification processes conducted by the institutions themselves or through suppliers themselves on behalf of the institution. The responsibility is also on large purchasers to ensure that these products come from suppliers whose compliance status is not in question. An approved suppliers list is maintained in this case.
Export Documentation Delays
There are delays in documentation for exports that are operational risks that can cause disruptions to the supply chain even if the quality of the product is satisfactory. Incomplete and conflicting documentation can cause delays in customs clearance, storage charges at the ports of entry, or the rejection of the Gauze Balls.
Typical difficulties include invoices being different, incorrect HS codes, certificates lacking details, or labeling inaccuracies. Such difficulties are further compounded for shipments involving more than one country, where the documentation needs to comply with both the exporting and importing nations’ requirements.
This risk is mitigated by institutions that partner with suppliers who have prior export performance. Pre-shipment document verification, document templates, and prior document sharing enable differences to be identified before leaving the factory. For NGOs contracted to operate within tight timelines, this preparation plays a crucial role in mission continuation.
Risk Mitigation Strategies
A successful risk management approach involves suppliers from the beginning and is present throughout the procurement process. Institutions that purchase Gauze Balls on a large scale do not purchase on the sole basis of price. A number of other factors have been considered.
Framework agreements and long-term contracts are usually deemed sufficient to create a certain degree of predictability regarding supply. These agreements establish criteria with respect to quality, auditing rights, and notification of change, which become standardized and follow a certain pattern if deviations occur through predefined escalation procedures.
Diversification is another approach. Certain institutions shortlist various manufacturers for Gauze Balls. In this way, reliance on a solitary supplier is avoided. Others keep buffer stock based on forecasted consumption to mitigate short-term challenges.
However, mitigation at this point is not about risk elimination. It is about risk reduction. Those institutions that monitor suppliers, are regularly reviewing documentation, and are in constant communication with suppliers are in better shape to mitigate risk before it materially impacts their clinical operations.
Why Risk Awareness Matters in Bulk Procurement
Procurement risks do not usually announce themselves explicitly. They build gradually and appear when the system is under stress. For Gauze Balls, which are used in various departments and settings, even slight disruptions could have far-reaching implications.
Risk-aware institutions view procurement not as a single-point activity or a one-time process or purchase. Instead, these institutions identify patterns and use structured risk mitigation strategies that safeguard not only patient outcomes but also efficiency.
In global healthcare supply chains, being reliable requires vision. One of the most useful actions that healthcare organizations should take in order to provide a guaranteed role for Gauze Balls in healthcare practice is to comprehend risks in healthcare purchases.
Why Global Buyers Choose BKA MED as a Gauze Ball Manufacturing Partner
In a world market full of players providing medical consumable products such as Gauze Balls for hospitals and procurement agencies, fewer manufacturers can genuinely be trusted by these institutions. BKA MED is among the reliable ones. This is because clients purchasing Gauze Balls from BKA MED not only look for cheap rates.

Here is a list detailing why BKA MED is a preferred manufacturer over its competition:
Manufacturing Scope & Vertical Integration
BKA MED has one of its greatest strengths in its vertically integrated manufacturing process. This means BKA MED does not contract third-party processors for the manufacturing of Gauze Balls. This includes cotton processing, gauze weaving, cutting, forming, and packaging. Sterilization is also done in-house.
Vertical integration enables reduced variability. It enables better control of absorbency, fibers, and product size. Such control for international consumers enables consistent quality delivery. Vertical integration also enables shorter lead times and reduced risk of reliance on dependencies, especially during peak seasons like emergencies and humanitarian crises.
Simply put, when fewer hands touch the product, fewer things go wrong.
Quality Systems & Compliance Strength
The quality systems in BKA MED are designed for global markets and not only for local distribution. The company has processes that work in line with ISO 13485 to ensure there is control over materials, production, inspection, and finished goods for release.
For Gauze Balls, this entails traceability of batches, verification of operation, and scaling of change. Whenever there is a change made in materials or process, this is evaluated and planned for change approval.
Consumers appreciate this rigor for the reason that the unpleasant surprise factor is avoided or minimized. With expectations clarified and fulfilled, risks are also minimized in the future. It is for these reasons, among others, that BKA MED establishes long-term contracts instead of transactional business with its suppliers.
Hospital and NGO Supply Experience
More than manufacturing capacity is required to supply Gauze Balls to hospitals and NGOs; what is needed is an understanding of how products are used in the real world. BKA MED has considerable experience supplying a wide variety of public hospitals, private healthcare networks, and international NGOs.
The experience shapes product design, packaging formats, and the nature of documentation. For example, shipments into NGOs require special labeling, tender documentation, and compliance declarations. Consistency and uninterrupted supply remain the key issues in hospital systems.
Because BKA MED understands these operational realities, buyers spend less time explaining requirements and more time focusing on deployment.
Customization & Scalability
Rarely do global buyers look for off-the-shelf solutions. BKA MED offers flexibility in customization related to Gauze Balls for size variations, number of ply, sterile or non-sterile format, private labeling, and bulk packaging configuration.
Of key importance is scalability: Whether the order is for a trial shipment or a multi-container contract, production capacity is designed to scale without ever compromising quality. This is exceptionally important for NGOs and centralized procurement agencies experiencing fluctuating demand.
Customization at scale can also mean standardization of products across regions for buyers, with necessary modifications to meet local regulatory or clinical requirements.
Export Documentation Readiness
The readiness of export is mostly taken for granted until a shipment is delayed. BKA MED treats documentation as part of the product, not an afterthought. Commercial invoices, packing lists, certificates of origin, test reports, and regulatory declarations are prepared accurately and consistently.
For Gauze Balls shipped to different regions, documentation is prepared in accordance with destination-specific requirements. This minimizes delays in customs, cost accumulation in storage, and administrative back-and-forth.
Buyers appreciate working with a manufacturer who can appreciate international logistics. When the documentation flows effortlessly, the supply chains remain stable.
A Partner, Not Just a Supplier
What truly gives BKA MED a competitive edge is its approach to and attitude. The company is positioned not as a material supplier or a potential customer but rather as a production partner.
For people who purchase Gauze Balls all over the world, this is an easier approach, as there is less friction, uncertainty, and risk involved. There is timely delivery of purchases. There is no variation in the product specifications
In an industry that values reliability much more than brands, BKA MED has built its reputation through its flawless delivery of the basics, time and again.
If your organization is in the process of evaluating its medical consumables, we would be pleased to assist. Hospitals and NGOs are welcome to contact us or request a quotation for gauze balls as needed. Our team supports buyers with both technical insight and flexible shipping options, helping ensure reliable stock levels across care facilities.

